Anti-High Mobility Group Box-1 Monoclonal Antibody Attenuates Seizure-Induced Cognitive Decline by Suppressing Neuroinflammation in an Adult Zebrafish Model
- PMID: 33732146
- PMCID: PMC7957017
- DOI: 10.3389/fphar.2020.613009
Anti-High Mobility Group Box-1 Monoclonal Antibody Attenuates Seizure-Induced Cognitive Decline by Suppressing Neuroinflammation in an Adult Zebrafish Model
Abstract
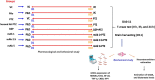
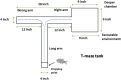
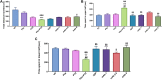
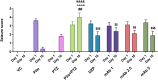
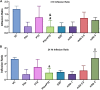
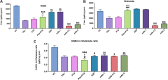
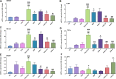
Epilepsy is a chronic brain disease afflicting around 70 million global population and is characterized by persisting predisposition to generate epileptic seizures. The precise understanding of the etiopathology of seizure generation is still elusive, however, brain inflammation is considered as a major contributor to epileptogenesis. HMGB1 protein being an initiator and crucial contributor of inflammation is known to contribute significantly to seizure generation via activating its principal receptors namely RAGE and TLR4 reflecting a potential therapeutic target. Herein, we evaluated an anti-seizure and memory ameliorating potential of an anti-HMGB1 monoclonal antibody (mAb) (1, 2.5 and 5 mg/kg, I.P.) in a second hit Pentylenetetrazol (PTZ) (80 mg/kg, I.P.) induced seizure model earlier stimulated with Pilocarpine (400 mg/kg, I.P.) in adult zebrafish. Pre-treatment with anti-HMGB1 mAb dose-dependently lowered the second hit PTZ-induced seizure but does not alter the disease progression. Moreover, anti-HMGB1 mAb also attenuated the second hit Pentylenetetrazol induced memory impairment in adult zebrafish as evidenced by an increased inflection ration at 3 and 24 h trail in T-maze test. Besides, decreased level of GABA and an upregulated Glutamate level was observed in the second hit PTZ induced group, which was modulated by pre-treatment with anti-HMGB1 mAb. Inflammatory responses occurred during the progression of seizures as evidenced by upregulated mRNA expression of HMGB1, TLR4, NF-κB, and TNF-α, in a second hit PTZ group, which was in-turn downregulated upon pre-treatment with anti-HMGB1 mAb reflecting its anti-inflammatory potential. Anti-HMGB1 mAb modulates second hit PTZ induced changes in mRNA expression of CREB-1 and NPY. Our findings indicates anti-HMGB1 mAb attenuates second hit PTZ-induced seizures, ameliorates related memory impairment, and downregulates the seizure induced upregulation of inflammatory markers to possibly protect the zebrafish from the incidence of further seizures through via modulation of neuroinflammatory pathway.
Keywords: anti-HMGB1 mAb; cognitive decline; epilepsy; neuroinflammation; seizures; zebrafish.
Copyright © 2021 Paudel, Othman and Shaikh.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Amini E., Golpich M., Farjam A. S., Kamalidehghan B., Mohamed Z., Ibrahim N. M., et al. (2018). Brain lipopolysaccharide preconditioning-induced gene reprogramming mediates a tolerance state in electroconvulsive shock model of epilepsy. Front. Pharmacol. 9, 416. 10.3389/fphar.2018.00416 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

