MSK1 downregulation is involved in inflammatory responses following subarachnoid hemorrhage in rats
- PMID: 33732337
- PMCID: PMC7903447
- DOI: 10.3892/etm.2021.9795
MSK1 downregulation is involved in inflammatory responses following subarachnoid hemorrhage in rats
Abstract
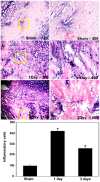
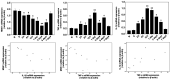
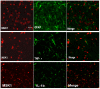
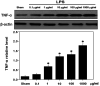
Subarachnoid hemorrhage (SAH) is a life-threatening neurological disease. Recently, inflammatory factors have been confirmed to be responsible for the brain damage associated with SAH. Therefore, studying the post-SAH inflammatory reaction may clarify the mechanism of SAH. Mitogen and stress-activated protein kinase 1 (MSK1) causes the phosphorylation of NF-κB and regulates the activity of pro-inflammatory transcription factors. The present study aimed to identify the potential role of MSK1 in inflammation and brain damage development following SAH. A cisterna magna blood injection model was established in Sprague-Dawley rats. Hematoxylin and eosin staining, reverse transcription-quantitative polymerase chain reaction assays and double immunofluorescence staining were used to analyze the role of MSK1, IL-1β and TNF-α in the inflammatory process after SAH. In a model of lipopolysaccharide-induced astrocyte inflammation, the effect of overexpressing MSK1 overexpression was analyzed by western blot analysis. The results demonstrated that MSK1 expression were negatively correlated with TNF-α and IL-1β expression levels, and reached peak levels 2 days after TNF-α and IL-1β. The double immunofluorescence staining results showed that the expression of MSK1 was in the same plane of view as TNF-α and IL-1β in the brain cortex. Furthermore, the in vitro studies indicated that the overexpression of MSK1 inhibited the expression of TNF-α and IL-1β following LPS challenge. These results imply that MSK1 may be involved in the inflammatory reaction following SAH, and may potentially serve as a negative regulator of inflammation.
Keywords: inflammation; mitogen and stress-activated protein kinase 1; subarachnoid hemorrhage.
Copyright: © Ning et al.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


References
-
- Connolly ES Jr, Rabinstein AA, Carhuapoma JR, Derdeyn CP, Dion J, Higashida RT, Hoh BL, Kirkness CJ, Naidech AM, Ogilvy CS, et al. American Heart Association Stroke Council; Council on Cardiovascular Radiology and Intervention; Council on Cardiovascular Nursing; Council on Cardiovascular Surgery and Anesthesia; Council on Clinical Cardiology: Guidelines for the management of aneurysmal subarachnoid hemorrhage: A guideline for healthcare professionals from the American Heart Association/american Stroke Association. Stroke. 2012;43:1711–1737. doi: 10.1161/STR.0b013e3182587839. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
