Paclitaxel increases axonal localization and vesicular trafficking of Nav1.7
- PMID: 33734317
- PMCID: PMC8320304
- DOI: 10.1093/brain/awab113
Paclitaxel increases axonal localization and vesicular trafficking of Nav1.7
Abstract
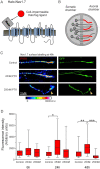
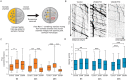
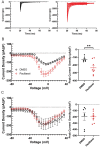
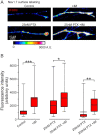
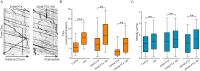
The microtubule-stabilizing chemotherapy drug paclitaxel (PTX) causes dose-limiting chemotherapy-induced peripheral neuropathy (CIPN), which is often accompanied by pain. Among the multifaceted effects of PTX is an increased expression of sodium channel Nav1.7 in rat and human sensory neurons, enhancing their excitability. However, the mechanisms underlying this increased Nav1.7 expression have not been explored, and the effects of PTX treatment on the dynamics of trafficking and localization of Nav1.7 channels in sensory axons have not been possible to investigate to date. In this study we used a recently developed live imaging approach that allows visualization of Nav1.7 surface channels and long-distance axonal vesicular transport in sensory neurons to fill this basic knowledge gap. We demonstrate concentration and time-dependent effects of PTX on vesicular trafficking and membrane localization of Nav1.7 in real-time in sensory axons. Low concentrations of PTX increase surface channel expression and vesicular flux (number of vesicles per axon). By contrast, treatment with a higher concentration of PTX decreases vesicular flux. Interestingly, vesicular velocity is increased for both concentrations of PTX. Treatment with PTX increased levels of endogenous Nav1.7 mRNA and current density in dorsal root ganglion neurons. However, the current produced by transfection of dorsal root ganglion neurons with Halo-tag Nav1.7 was not increased after exposure to PTX. Taken together, this suggests that the increased trafficking and surface localization of Halo-Nav1.7 that we observed by live imaging in transfected dorsal root ganglion neurons after treatment with PTX might be independent of an increased pool of Nav1.7 channels. After exposure to inflammatory mediators to mimic the inflammatory condition seen during chemotherapy, both Nav1.7 surface levels and vesicular transport are increased for both low and high concentrations of PTX. Overall, our results show that PTX treatment increases levels of functional endogenous Nav1.7 channels in dorsal root ganglion neurons and enhances trafficking and surface distribution of Nav1.7 in sensory axons, with outcomes that depend on the presence of an inflammatory milieu, providing a mechanistic explanation for increased excitability of primary afferents and pain in CIPN.
Keywords: Nav1.7; chemotherapy-induced peripheral neuropathy; live microscopy; paclitaxel; peripheral pain.
© The Author(s) (2021). Published by Oxford University Press on behalf of the Guarantors of Brain.
Figures

Comment in
-
Uncomfortably numb: how Nav1.7 mediates paclitaxel-induced peripheral neuropathy.Brain. 2021 Jul 28;144(6):1621-1623. doi: 10.1093/brain/awab196. Brain. 2021. PMID: 34009327 Free PMC article.
References
-
- Flatters SJL, Dougherty PM, Colvin LA.. Clinical and preclinical perspectives on chemotherapy-induced peripheral neuropathy (CIPN): A narrative review. Br J Anaesth. 2017;119(4):737–749. - PubMed
-
- Gornstein E, Schwarz TL.. The paradox of paclitaxel neurotoxicity: Mechanisms and unanswered questions. Neuropharmacology. 2014;76 Pt A:175–183. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

