Deletion of arginase 2 attenuates neuroinflammation in an experimental model of optic neuritis
- PMID: 33735314
- PMCID: PMC7971528
- DOI: 10.1371/journal.pone.0247901
Deletion of arginase 2 attenuates neuroinflammation in an experimental model of optic neuritis
Abstract
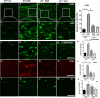
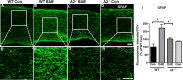
Vision impairment due to optic neuritis (ON) is one of the major clinical presentations in Multiple Sclerosis (MS) and is characterized by inflammation and degeneration of the optic nerve and retina. Currently available treatments are only partially effective and have a limited impact on the neuroinflammatory pathology of the disease. A recent study from our laboratory highlighted the beneficial effect of arginase 2 (A2) deletion in suppressing retinal neurodegeneration and inflammation in an experimental model of MS. Utilizing the same model, the present study investigated the impact of A2 deficiency on MS-induced optic neuritis. Experimental autoimmune encephalomyelitis (EAE) was induced in wild-type (WT) and A2 knockout (A2-/-) mice. EAE-induced cellular infiltration, as well as activation of microglia and macrophages, were reduced in A2-/- optic nerves. Axonal degeneration and demyelination seen in EAE optic nerves were observed to be reduced with A2 deletion. Further, the lack of A2 significantly ameliorated astrogliosis induced by EAE. In conclusion, our findings demonstrate a critical involvement of arginase 2 in mediating neuroinflammation in optic neuritis and suggest the potential of A2 blockade as a targeted therapy for MS-induced optic neuritis.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures




References
-
- Zwibel HL, Smrtka J. Improving quality of life in multiple sclerosis: an unmet need. Am J Manag Care. 2011;17 Suppl 5 Improving:S139–45. Epub 2011/07/27. . - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

