The adhesion GPCR Adgrg6 (Gpr126): Insights from the zebrafish model
- PMID: 33735533
- PMCID: PMC11475505
- DOI: 10.1002/dvg.23417
The adhesion GPCR Adgrg6 (Gpr126): Insights from the zebrafish model
Abstract
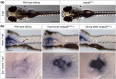
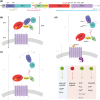
Adhesion GPCRs are important regulators of conserved developmental processes and represent an untapped pool of potential targets for drug discovery. The adhesion GPCR Adgrg6 (Gpr126) has critical developmental roles in Schwann cell maturation and inner ear morphogenesis in the zebrafish embryo. Mutations in the human ADGRG6 gene can result in severe deficits in peripheral myelination, and variants have been associated with many other disease conditions. Here, we review work on the zebrafish Adgrg6 signaling pathway and its potential as a disease model. Recent advances have been made in the analysis of the structure of the Adgrg6 receptor, demonstrating alternative structural conformations and the presence of a conserved calcium-binding site within the CUB domain of the extracellular region that is critical for receptor function. Homozygous zebrafish adgrg6 hypomorphic mutants have been used successfully as a whole-animal screening platform, identifying candidate molecules that can influence signaling activity and rescue mutant phenotypes. These compounds offer promise for further development as small molecule modulators of Adgrg6 pathway activity.
Keywords: Adgrg6; Gpr126; aGPCR; drug screening; heart; inner ear; myelination; skeleton; zebrafish.
© 2021 The Authors. genesis published by Wiley Periodicals LLC.
Conflict of interest statement
GRW is an employee and shareholder of Sosei Heptares. The other authors declare no competing interests.
Figures

References
-
- Beliu, G. , Altrichter, S. , Guixà‐González, R. , Hemberger, M. , Brauer, I. , Dahse, A.‐K. , … Langenhan, T. (2021). Tethered agonist exposure in intact adhesion/class B2 GPCRs through intrinsic structural flexibility of the GAIN domain. Molecular Cell, 81, 905–921. 10.1016/j.molcel.2020.12.042 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

