Role of the superior salivatory nucleus in parasympathetic control of choroidal blood flow and in maintenance of retinal health
- PMID: 33736985
- PMCID: PMC8087653
- DOI: 10.1016/j.exer.2021.108541
Role of the superior salivatory nucleus in parasympathetic control of choroidal blood flow and in maintenance of retinal health
Abstract
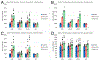
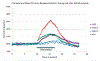
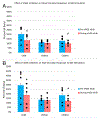
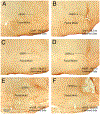
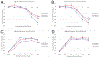
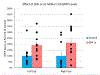
The vasodilatory pterygopalatine ganglion (PPG) innervation of the choroid is under the control of preganglionic input from the superior salivatory nucleus (SSN), the parasympathetic portion of the facial motor nucleus. We sought to confirm that choroidal SSN drives a choroid-wide vasodilation and determine if such control is important for retinal health. To the former end, we found, using transscleral laser Doppler flowmetry, that electrical activation of choroidal SSN significantly increased choroidal blood flow (ChBF), at a variety of choroidal sites that included more posterior as well as more anterior ones. We further found that the increases in ChBF were significantly reduced by inhibition of neuronal nitric oxide synthase (nNOS), thus implicating nitrergic PPG terminals in the SSN-elicited ChBF increases. To evaluate the role of parasympathetic control of ChBF in maintaining retinal health, some rats received unilateral lesions of SSN, and were evaluated functionally and histologically. In eyes ipsilateral to choroidal SSN destruction, we found that the flash-evoked scotopic electroretinogram a-wave and b-wave peak amplitudes were both significantly reduced by 10 weeks post lesion. Choroidal baroregulation was evaluated in some of these rats, and found to be impaired in the low systemic arterial blood pressure (ABP) range where vasodilation normally serves to maintain stable ChBF. In retina ipsilateral to SSN destruction, the abundance of Müller cell processes immunolabeled for glial fibrillary acidic protein (GFAP) and GFAP message were significantly upregulated. Our studies indicate that the SSN-PPG circuit mediates parasympathetic vasodilation of choroid, which appears to contribute to ChBF baroregulation during low ABP. Our results further indicate that impairment in this adaptive mechanism results in retinal dysfunction and pathology within months of the ChBF disturbance, indicating its importance for retinal health.
Keywords: Autonomic; Choroidal blood flow (ChBF); Parasympathetic; Retinal degeneration; Superior salivatory nucleus (SSN).
Copyright © 2021 The Authors. Published by Elsevier Ltd.. All rights reserved.
Conflict of interest statement
Declarations of competing interest
None.
Figures



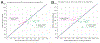


References
-
- Alm A, 1992. Ocular circulation. In: Hart WM (Ed.), Adler’s Physiology of the Eye: Clinical Application. Mosby, St Louis, pp. 198–227.
-
- Bill A, 1985. Some aspects of the ocular circulation. Invest. Ophthalmol. Vis. Sci 26, 410–424. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

