Adult-born neurons immature during learning are necessary for remote memory reconsolidation in rats
- PMID: 33741954
- PMCID: PMC7979763
- DOI: 10.1038/s41467-021-22069-4
Adult-born neurons immature during learning are necessary for remote memory reconsolidation in rats
Abstract
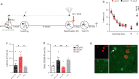
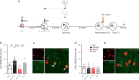
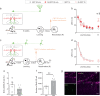
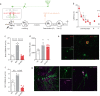
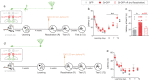
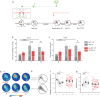
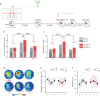
Memory reconsolidation, the process by which memories are again stabilized after being reactivated, has strengthened the idea that memory stabilization is a highly plastic process. To date, the molecular and cellular bases of reconsolidation have been extensively investigated particularly within the hippocampus. However, the role of adult neurogenesis in memory reconsolidation is unclear. Here, we combined functional imaging, retroviral and chemogenetic approaches in rats to tag and manipulate different populations of rat adult-born neurons. We find that both mature and immature adult-born neurons are activated by remote memory retrieval. However, only specific silencing of the adult-born neurons immature during learning impairs remote memory retrieval-induced reconsolidation. Hence, our findings show that adult-born neurons immature during learning are required for the maintenance and update of remote memory reconsolidation.
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Postretrieval Relearning Strengthens Hippocampal Memories via Destabilization and Reconsolidation.J Neurosci. 2019 Feb 6;39(6):1109-1118. doi: 10.1523/JNEUROSCI.2618-18.2018. Epub 2018 Dec 26. J Neurosci. 2019. PMID: 30587543 Free PMC article.
-
Reconstructing a new hippocampal engram for systems reconsolidation and remote memory updating.Neuron. 2025 Feb 5;113(3):471-485.e6. doi: 10.1016/j.neuron.2024.11.010. Epub 2024 Dec 16. Neuron. 2025. PMID: 39689709
-
Recognition memory reconsolidation requires hippocampal Zif268.Sci Rep. 2019 Nov 12;9(1):16620. doi: 10.1038/s41598-019-53005-8. Sci Rep. 2019. PMID: 31719567 Free PMC article.
-
Determinants to trigger memory reconsolidation: The role of retrieval and updating information.Neurobiol Learn Mem. 2017 Jul;142(Pt A):4-12. doi: 10.1016/j.nlm.2016.12.005. Epub 2016 Dec 21. Neurobiol Learn Mem. 2017. PMID: 28011191 Review.
-
[Consolidation, reactivation and reconsolidation of memory].Zh Vyssh Nerv Deiat Im I P Pavlova. 2014 Mar-Apr;64(2):123-36. Zh Vyssh Nerv Deiat Im I P Pavlova. 2014. PMID: 25713863 Review. Russian.
Cited by
-
Changes in the dopaminergic circuitry and adult neurogenesis linked to reinforcement learning in corvids.Front Neurosci. 2024 May 14;18:1359874. doi: 10.3389/fnins.2024.1359874. eCollection 2024. Front Neurosci. 2024. PMID: 38808028 Free PMC article.
-
What Is Adult Hippocampal Neurogenesis Good for?Front Neurosci. 2022 Apr 15;16:852680. doi: 10.3389/fnins.2022.852680. eCollection 2022. Front Neurosci. 2022. PMID: 35495058 Free PMC article.
-
Regulation of neural stem cells by innervating neurons.J Neurochem. 2025 Jan;169(1):e16287. doi: 10.1111/jnc.16287. J Neurochem. 2025. PMID: 39775528 Free PMC article. Review.
-
Hypothalamic modulation of adult hippocampal neurogenesis in mice confers activity-dependent regulation of memory and anxiety-like behavior.Nat Neurosci. 2022 May;25(5):630-645. doi: 10.1038/s41593-022-01065-x. Epub 2022 May 6. Nat Neurosci. 2022. PMID: 35524139 Free PMC article.
-
Age-related decline in cognitive flexibility is associated with the levels of hippocampal neurogenesis.Front Neurosci. 2023 Aug 14;17:1232670. doi: 10.3389/fnins.2023.1232670. eCollection 2023. Front Neurosci. 2023. PMID: 37645372 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

