Systematic review of outcome domains and instruments used in designs of clinical trials for interventions that seek to restore bilateral and binaural hearing in adults with unilateral severe to profound sensorineural hearing loss ('single-sided deafness')
- PMID: 33743802
- PMCID: PMC7981927
- DOI: 10.1186/s13063-021-05160-5
Systematic review of outcome domains and instruments used in designs of clinical trials for interventions that seek to restore bilateral and binaural hearing in adults with unilateral severe to profound sensorineural hearing loss ('single-sided deafness')
Abstract
Background: This systematic review aimed to identify, compare and contrast outcome domains and outcome instruments reported in studies investigating interventions that seek to restore bilateral (two-sided) and/or binaural (both ears) hearing in adults with single-sided deafness (SSD). Findings can inform the development of evidence-based guidance to facilitate design decisions for confirmatory trials.
Methods: Records were identified by searching MEDLINE, EMBASE, PubMed, CINAHL, ClinicalTrials.gov, ISRCTN, CENTRAL, WHO ICTRP and the NIHR UK clinical trials gateway. The search included records published from 1946 to March 2020. Included studies were those as follows: (a) recruiting adults aged 18 years or older diagnosed with SSD of average threshold severity worse than 70 dB HL in the worse-hearing ear and normal (or near-normal) hearing in the better-hearing ear, (b) evaluating interventions to restore bilateral and/or binaural hearing and (c) enrolling those adults in a controlled trial, before-and-after study or cross-over study. Studies that fell just short of the participant eligibility criteria were included in a separate sensitivity analysis.
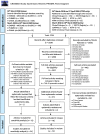
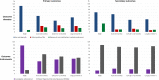
Results: Ninety-six studies were included (72 full inclusion, 24 sensitivity analysis). For fully included studies, 37 exclusively evaluated interventions to re-establish bilateral hearing and 29 exclusively evaluated interventions to restore binaural hearing. Overall, 520 outcome domains were identified (350 primary and 170 secondary). Speech-related outcome domains were the most common (74% of studies), followed by spatial-related domains (60% of studies). A total of 344 unique outcome instruments were reported. Speech-related outcome domains were measured by 73 different instruments and spatial-related domains by 43 different instruments. There was considerable variability in duration of follow-up, ranging from acute (baseline) testing to 10 years after the intervention. The sensitivity analysis identified no additional outcome domains.
Conclusions: This review identified large variability in the reporting of outcome domains and instruments in studies evaluating the therapeutic benefits and harms of SSD interventions. Reports frequently omitted information on what domains the study intended to assess, and on what instruments were used to measure which domains.
Trial registration: The systematic review protocol is registered on PROSPERO (International Prospective Register of Systematic Reviews): Registration Number CRD42018084274 . Registered on 13 March 2018, last revised on 7th of May 2019.
Keywords: Clinical trial design; Hearing interventions; Measurement instruments; Outcome domains; Single-sided deafness.
Conflict of interest statement
PTK declared research grants and support in kind awarded to his institution for research projects in the field of SSD from manufacturers of hearing devices. All other authors declare that they have no competing interests.
Figures
References
-
- Van de Heyning P, Távora-Vieira D, Mertens G, Van Rompaey V, Rajan GP, Müller J, Hempel JM, Leander D, Polterauer D, Marx M, Usami SI, Kitoh R, Miyagawa M, Moteki H, Smilsky K, Baumgartner WD, Keintzel TG, Sprinzl GM, Wolf-Magele A, et al. Towards a unified testing framework for single-sided deafness studies: A consensus paper. Audiol Neurootol. 2017;21:391–398. doi: 10.1159/000455058. - DOI - PMC - PubMed
-
- Widen JE, Folsom RC, Cone-Wesson B, Carty L, Dunnell JJ, Koebsell K, Levi A, Mancl L, Ohlrich B, Trouba S, Gorga MP, Sininger YS, Vohr BR, Norton SJ. Identification of neonatal hearing impairment: hearing status at 8 to 12 months corrected age using a visual reinforcement audiometry protocol. Ear Hear. 2000;21:471–487. doi: 10.1097/00003446-200010000-00011. - DOI - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

