Efficacy of a Coriolus versicolor-Based Vaginal Gel in Women With Human Papillomavirus-Dependent Cervical Lesions: The PALOMA Study
- PMID: 33746195
- PMCID: PMC7984756
- DOI: 10.1097/LGT.0000000000000596
Efficacy of a Coriolus versicolor-Based Vaginal Gel in Women With Human Papillomavirus-Dependent Cervical Lesions: The PALOMA Study
Abstract
Objective: The aim of the study was to evaluate the efficacy of Papilocare, a Coriolus versicolor-based vaginal gel, in repairing human papillomavirus (HPV)-related low-grade cervical lesions.
Methods: The study is a multicenter, open-label, randomized, parallel-group, watchful waiting approach-controlled trial involving 91 HPV-positive women with low-grade Pap smear alterations and consistent colposcopy.
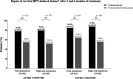
Results: The percentage of patients with normal Pap smear and concordant colposcopy 3 and 6 months after receiving treatment (78.0% and 84.9%) was significantly higher than without treatment (54.8% and 64.5%), especially in high-risk HPV patients (79.5% and 87.8% vs 52.0% and 56.0%). At 6-month visit, overall HPV clearance was achieved by a greater number of patients receiving treatment (59.6%) compared with those without treatment (41.9%), especially high-risk HPV ones (62.5% vs 40.0%). The cervical re-epithelization score was significantly higher with treatment (mean = 4.5) than without (mean = 4.1). Compared with baseline, perceived stress decreased in the treatment group (from 21.1 to 19.0) and increased in the control group (from 17.7 to 20.7). A total of 7 possible or probable treatment-related adverse events were reported, most of them (n = 6) being mild or moderate in severity.
Conclusions: Treatment with Papilocare has demonstrated a better clinical benefit than the conventional watchful waiting approach in clinical practice for total and high-risk HPV patients in terms of its efficacy to treat HPV-related cervical lesions and to clear all HPV strains after a single 6-month period. It has demonstrated an adequate safety and tolerability and confers additional benefits such as higher re-epithelization, stress reduction, and high treatment adherence.
Copyright © 2021 The Author(s). Published by Wolters Kluwer Health, Inc. on behalf of the ASCCP.
Conflict of interest statement
J.C. declares that he has received travel and/or research grants and/or honoraria for conferences and/or consultancy fees from Genómica, GSK, Merck, Procare Health, Qiagen, Roche, and SPMSD. S.P. declares that he has a financial relationship (as a scientific advisor, member of the advisory board, or consultant) with Pfizer, Amgen, MSD, Sandoz, Procare Health, Bayer, MSD, Serelys, and Shionogi. Furthermore, as a speaker or member of the advisory board, he has received research grants or honoraria from Servier, Pfizer, Abbott, Shionogi, Amgen, Novo Nordisk, Theramex, Bayer Healthcare, Serelys, and Gedeon Richter. D.D. declares that he has received speaking and consultancy fees from Sanofi Pasteur, MSD, and Procare Health. L.S. declares that he has received speaking and consultancy fees from Shionogi, Iprad, and Procare Health. A.C.L. declares that he has received consultancy fees from Procare Health. S.P.G. declares that she has received travel and/or research grants and/or honoraria for conferences and/or scientific advisory and consultancy fees from Isdín, Pfizer, Servier, Amgen, MSD, Kern-Gynea, Casen Recordati, Sandoz, Procare Health, Bayer, Lacer, Shionogi, GSK, Bioiberica, Theramex, Gedeon Richter, Iprad, Seid, Ordesa, and Zambon. C.V. declares that she has received speaking fees from Procare Health. C.C.-M. declares that she has received speaking fees from Bayer and a grant for congress attendance from GSK. P.C. declares that he has received travel grants, conference registration grants and/or honoraria for conferences, and/or consultancy fees from Genómica, GSK, MSD, Abex, Shionogi, Amgen, Lacer, and Pfizer. J.d.l.F. declares that he has received travel and/or research grants and/or honoraria for conferences and/or consultancy fees from GSK, Merck, Procare Health, Roche, SPMSD, Italfármaco, Adamed, Pfizer, and Effik. J.A.L. declares that he has received honoraria for lectures from Procare Health, Gedeon Richter, and Sanofi. The other authors have declared they have no conflicts of interest.
Figures


Comment in
-
Cytology Is Not Reliable to Evaluate Cervical Disease Regression.J Low Genit Tract Dis. 2021 Oct 1;25(4):328. doi: 10.1097/LGT.0000000000000615. J Low Genit Tract Dis. 2021. PMID: 34265819 No abstract available.
-
Concordant Sum of Cytology and Colposcopy Is Validated by Guidelines to Evaluate the Evolution of Low-Grade Lesions.J Low Genit Tract Dis. 2021 Oct 1;25(4):329. doi: 10.1097/LGT.0000000000000616. J Low Genit Tract Dis. 2021. PMID: 34265820 No abstract available.
References
-
- World Health Organization . Human papillomavirus (HPV) and cervical cancer. Available at: https://www.who.int/news-room/fact-sheets/detail/human-papillomavirus-(h.... Accessed May 21, 2020.
-
- Ho GY Bierman R Beardsley L, et al. . Natural history of cervicovaginal papillomavirus infection in young women. N Engl J Med 1998;338:423–8. - PubMed
-
- McBride E Tatar O Rosberger Z, et al. . Emotional response to testing positive for human papillomavirus at cervical cancer screening: a mixed method systematic review with meta-analysis. Health Psychol Rev 2020;1–35. - PubMed
-
- Thangarajah F Einzmann T Bergauer F, et al. . Cervical screening program and the psychological impact of an abnormal Pap smear: a self-assessment questionnaire study of 590 patients. Arch Gynecol Obstet 2016;293:391–8. - PubMed
-
- Need for safe, non-surgical treatment for clearing human papilloma virus (HPV). Available at : https://livbio.co.uk/need-for-safe-non-surgical-treatment-for-clearing-h.... Accessed September 17, 2020.
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

