A 9-Month-Old with Skeletal Abnormalities and a Consanguineous Sibling with Mucopolysaccharidosis IVA: The Role of Urinary Glycosaminoglycan Testing in Disease Diagnosis and Treatment Monitoring
- PMID: 33746520
- PMCID: PMC7940721
- DOI: 10.1177/1179547621999409
A 9-Month-Old with Skeletal Abnormalities and a Consanguineous Sibling with Mucopolysaccharidosis IVA: The Role of Urinary Glycosaminoglycan Testing in Disease Diagnosis and Treatment Monitoring
Abstract
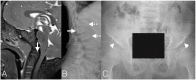
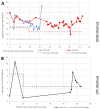
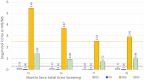
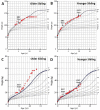
Mucopolysaccharidosis IVA (MPS IVA) is a rare autosomal recessive lysosomal storage disorder resulting from N-acetylgalactosamine-6-sulfatase (GALNS) deficiency that occurs in approximately 1 in 76 000 to 1 in 640 000 live births. Given that the diagnosis of MPS IVA relies heavily on the results of initial urine glycosaminoglycan (GAG) screening, cases that present with falsely normal urine GAG concentrations can delay the diagnosis and follow-up care for patients. This case study follows a patient diagnosed with MPS IVA at 9 months of age based on relation to a consanguineous 3-year-old sibling with MPS IVA and the use of direct enzyme activity analysis. Details regarding skeletal presentation and identification of genetic variants are presented along with data on follow-up urinary GAG monitoring during treatment with enzyme replacement therapy and treatment for a growth hormone disorder.
Keywords: Mucopolysaccharidosis IVA; N-acetylgalactosamine-6-sulfatase (GALNS); enzyme replacement therapy; glycosaminoglycan; growth hormone deficiency; keratan sulfate.
© The Author(s) 2021.
Conflict of interest statement
Declaration of conflicting interests:The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures
References
-
- Bunge S, Kleijer WJ, Tylki-Szymanska A, et al. Identification of 31 novel mutations in the N-acetylgalactosamine-6-sulfatase gene reveals excessive allelic heterogeneity among patients with Morquio A syndrome. Hum Mutat. 1997;10:223-232. doi: 10.1002/(SICI)1098-1004(1997)10:3<223::AID-HUMU8>3.0.CO;2-J - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

