Reporter gene assays for screening and identification of novel molting hormone- and juvenile hormone-like chemicals
- PMID: 33746544
- PMCID: PMC7953021
- DOI: 10.1584/jpestics.D20-079
Reporter gene assays for screening and identification of novel molting hormone- and juvenile hormone-like chemicals
Abstract
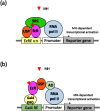
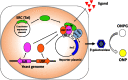
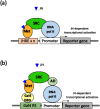
A reporter gene assay (RGA) is used to investigate the activity of synthetic chemicals mimicking the molting hormones (MHs) and juvenile hormones (JHs) of insects, so-called insect growth regulators (IGRs). The MH receptor, a heterodimer of the ecdysone receptor (EcR) and ultraspiracle (USP), and the JH receptor Methoprene-tolerant (Met) are ligand-dependent transcription factors. Ligand-bound EcR-USP and Met bind to specific cis-acting DNA elements, referred to as the ecdysone-responsive element (EcRE) and the JH-responsive element (JHRE), respectively, in order to transactivate target genes. Insect hormone-induced transactivation systems have been reconstituted by the introduction of reporter genes under the control of EcRE and JHRE, or two-hybrid reporter genes, into insect, mammalian, and yeast cells expressing receptor proteins. RGA is easy to use and convenient for examining the MH- and JH-like activities of synthetic chemicals and is suitable for the high-throughput screening of novel structural classes of chemicals targeting EcR-USP and Met.
Keywords: agonist; ecdysone receptor; insect growth regulator; juvenile hormone receptor; reporter gene assay.
© Pesticide Science Society of Japan 2021. This is an open access article distributed under the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International (CC BY-NC-ND 4.0) License (https://creativecommons.org/licenses/by-nc-nd/4.0/).
Figures

References
-
- T. S. Dhadialla, G. R. Carlson and D. P. Le: New insecticides with ecdysteroidal and juvenile hormone activity. Annu. Rev. Entomol. 43, 545–569 (1998). - PubMed
-
- H. Tunaz and N. Uygun: Insect growth regulators for insect pest control. Turk. J. Agric. For. 28, 377–387 (2004).
-
- H. C. Smith, C. K. Cavanaugh, J. L. Friz, C. S. Thompson, J. A. Saggers, E. L. Michelotti, J. Garcia and C. M. Tice: Synthesis and SAR of cis-1-benzoyl-1,2,3,4-tetrahydroquinoline ligands for control of gene expression in ecdysone responsive systems. Bioorg. Med. Chem. Lett. 13, 1943–1946 (2003). - PubMed
-
- S. R. Palli, C. M. Tice, V. M. Margam and A. M. Clark: Biochemical mode of action and differential activity of new ecdysone agonists against mosquitoes and moths. Arch. Insect Biochem. Physiol. 58, 234–242 (2005). - PubMed
-
- E. Becker: Über versuche zur anreicherung und physiologischer charakterisierung des wirkstoffes der puparisierung. Biol. Zbl. 61, 360–388 (1941).
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

