One-step vapor deposition of fluorinated polycationic coating to fabricate antifouling and anti-infective textile against drug-resistant bacteria and viruses
- PMID: 33746567
- PMCID: PMC7962519
- DOI: 10.1016/j.cej.2021.129368
One-step vapor deposition of fluorinated polycationic coating to fabricate antifouling and anti-infective textile against drug-resistant bacteria and viruses
Abstract
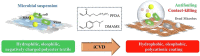
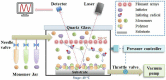
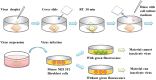
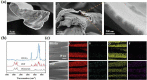
The ongoing pandemic caused by the novel coronavirus has turned out to be one of the biggest threats to the world, and the increase of drug-resistant bacterial strains also threatens the human health. Hence, there is an urgent need to develop novel anti-infective materials with broad-spectrum anti-pathogenic activity. In the present study, a fluorinated polycationic coating was synthesized on a hydrophilic and negatively charged polyester textile via one-step initiated chemical vapor deposition of poly(dimethyl amino methyl styrene-co-1H,1H,2H,2H-perfluorodecyl acrylate) (P(DMAMS-co-PFDA), PDP). The surface characterization results of SEM, FTIR, and EDX demonstrated the successful synthesis of PDP coating. Contact angle analysis revealed that PDP coating endowed the polyester textile with the hydrophobicity against the attachment of different aqueous foulants such as blood, coffee, and milk, as well as the oleophobicity against paraffin oil. Zeta potential analysis demonstrated that the PDP coating enabled a transformation of negative charge to positive charge on the surface of polyester textile. The PDP coating exhibited excellent contact-killing activity against both gram-negative Escherichia coli and gram-positive methicillin-resistant Staphylococcus aureus, with the killing efficiency of approximate 99.9%. In addition, the antiviral capacity of PDP was determined by a green fluorescence protein (GFP) expression-based method using lentivirus-EGFP as a virus model. The PDP coating inactivated the negatively charged lentivirus-EGFP effectively. Moreover, the coating showed good biocompatibility toward mouse NIH 3T3 fibroblast cells. All the above properties demonstrated that PDP would be a promising anti-pathogenic polymeric coating with wide applications in medicine, hygiene, hospital, etc., to control the bacterial and viral transmission and infection.
Keywords: Antibacterial; Antifouling; Antiviral; Fluorinated hydrophobic polymer; Initiated chemical vapor deposition (iCVD); Polycationic coating.
© 2021 Elsevier B.V. All rights reserved.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures




References
-
- Magiorakos A.P., Srinivasan A., Carey R.B., Carmeli Y., Falagas M.E., Giske C.G., Harbarth S., Hindler J.F., Kahlmeter G., Olsson-Liljequist B., Paterson D.L., Rice L.B., Stelling J., Struelens M.J., Vatopoulos A., Weber J.T., Monnet D.L. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin. Microbiol. Infect. 2012;18(3):268–281. doi: 10.1111/j.1469-0691.2011.03570.x. - DOI - PubMed
-
- Li R., Dou J., Jiang Q., Li J., Xie Z., Liang J., Ren X. Preparation and antimicrobial activity of β-cyclodextrin derivative copolymers/cellulose acetate nanofibers. Chem. Eng. J. 2014;248:264–272. doi: 10.1016/j.cej.2014.03.042. - DOI
LinkOut - more resources
Full Text Sources
Other Literature Sources
