Compliance of Published Randomized Controlled Trials on the Effect of Physical Activity on Primary Dysmenorrhea with the Consortium's Integrated Report on Clinical Trials Statement: A Critical Appraisal of the Literature
- PMID: 33747832
- PMCID: PMC7968594
- DOI: 10.4103/ijnmr.IJNMR_223_19
Compliance of Published Randomized Controlled Trials on the Effect of Physical Activity on Primary Dysmenorrhea with the Consortium's Integrated Report on Clinical Trials Statement: A Critical Appraisal of the Literature
Abstract
Background: Randomized Controlled Trials (RCTs) are reliable methods for the evaluation of treatment effectiveness, which should be rigorous and must report with clarity. This study aimed to assess the compliance of published RCTs about the effect of physical activity on primary dysmenorrhea with the CONSORT 2017 statement.
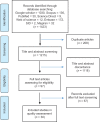
Materials and methods: In this study, the review of literature was carried out based on Consolidated Standards Of Reporting Trials (CONSORT). All the clinical trials focused on the effect of the physical activity on primary dysmenorrhea indexed in Web of Science, Pubmed, Scopus, Google Scholar, Science Direct, Embase, Magiran and Scientific Information Database (SID) were searched using keywords of dysmenorrhea, randomized clinical trial, physical activity and exercise from 2000 to 2019. Out of 1423 articles, 30 RCTs were critically appraised using CONSORT 2017 checklist. The reporting quality score of articles was identified between zero and 43.
Results: The compliance rate with the CONSORT checklist was 55.58%. The mean (SD) score of the reporting quality was 23.37 (-5.15) with a minimum of 16 and a maximum of 37. The maximum weakness was in reporting the sample size and full trial protocol 23.33% and 6.67% respectively. Regarding new items of the consort 2017, if the blinding was not possible, the description of any attempts to limit bias was not described in 70% of articles.
Conclusions: Reporting sample size, trial protocol, method of blinding, and control of bias are issues that require more attention in reporting of RCT studies. We recommend that the authors use the CONSORT 2017 statement for conducting and reporting the clinical trials.
Keywords: Exersice; dysmenorrhea; randomized controlled trial; review.
Copyright: © 2020 Iranian Journal of Nursing and Midwifery Research.
Conflict of interest statement
Nothing to declare.
Figures
References
-
- Uetani K, Nakayama T, Ikai H, Yonemoto N, Moher D. Quality of reports on randomized controlled trials conducted in Japan: Evaluation of adherence to the CONSORT statement. Intern Med. 2009;48:307–13. - PubMed
-
- Shein-Chung C, Liu J. Design and Analysis of Clinical Trials: Concepts and Methodologies. Wiley. 2013
-
- Gagnier J, Boon H, Rochon P, Barnes J, Moher D, Bombardier C, et al. Improving the quality of reporting of randomized controlled trials evaluating herbal interventions: Implementing the CONSORT statement [corrected] Explore (NY) 2006;2:143–9. - PubMed
Publication types
LinkOut - more resources
Full Text Sources