Immune-Related Adverse Events and Their Association With the Effectiveness of PD-1/PD-L1 Inhibitors in Non-Small Cell Lung Cancer: A Real-World Study From China
- PMID: 33747922
- PMCID: PMC7973369
- DOI: 10.3389/fonc.2021.607531
Immune-Related Adverse Events and Their Association With the Effectiveness of PD-1/PD-L1 Inhibitors in Non-Small Cell Lung Cancer: A Real-World Study From China
Abstract
Background: Programmed cell death-1/programmed cell death ligand-1 (PD-1/PD-L1) inhibitors are increasingly used in China, but no real-world data are available about the immune-related adverse events (irAEs). This real-world retrospective study aimed to assess the safety and effectiveness of PD-1/PD-L1 inhibitors in patients with non-small cell lung cancer (NSCLC) and to analyze the association between irAEs and effectiveness.
Methods: This was a retrospective study of the clinical data of patients with NSCLC treated with PD-1/PD-L1 inhibitors from August 2016 to November 2019 at Beijing Cancer Hospital. The patients were divided into the irAE or non-irAE groups. Overall adverse events, the impact of irAE on tumor response, and the association of irAEs with effectiveness were evaluated.
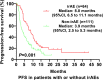
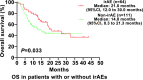
Results: One hundred and ninety-one patients were included, including 70 (36.6%) patients in the irAE group and 121 (63.4%) patients in the non-irAE group. AE, grades 3-5 AEs, and irAE occurred in 107 (56.0%), 24 (12.6%), and 70 (36.6%) of the patients, respectively. The objective response rate (ORR) and disease control rate (DCR) were higher in the irAE group compared with the non-irAE group (42.0% vs. 25.8%, P=0.038; 91.9% vs. 70.8%, P=0.002). Multivariable analyses identified that irAE were associated with progression-free survival (HR=0.62, 95%CI: 0.43-0.91; P=0.015), but not with overall survival (HR=0.76, 95%CI: 0.44-1.28; P=0.299).
Conclusion: In NSCLC treated with PD-1/PD-L1 inhibitors, patients with irAEs showed improved effectiveness over patients without irAEs. Future studies of anti-PD-1/PD-L1 immunotherapy should explore this association and the underlying biological mechanisms of efficacy.
Keywords: adverse events; immunotherapy; non-small cell lung cancer; objective response; real-world evidence; survival.
Copyright © 2021 Chen, Nie, Dai, Hu, Zhang, Han, Ma, Tian, Han, Wu, Wang, Long, Zhang and Fang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

