Selective inhibition of V600E-mutant BRAF gene induces apoptosis in thyroid carcinoma cell lines
- PMID: 33748026
- PMCID: PMC7943282
- DOI: 10.4174/astr.2021.100.3.127
Selective inhibition of V600E-mutant BRAF gene induces apoptosis in thyroid carcinoma cell lines
Abstract
Purpose: Papillary thyroid cancer (PTC) has a high incidence of BRAFV600E mutation. The purpose of this study was to evaluate the potential relationship between thyroiditis and BRAFV600E mutation status in patients with PTC. We investigated how a selective inhibitor of BRAFV600E PLX4032 affects the proliferation and inflammatory cytokine levels of thyroid cancer.
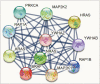
Methods: Two thyroid cancer cell lines TPC1 and 8505C were treated with PLX4032, an analysis was done on cell growth, cell cycle, the degree of apoptosis, and levels of inflammatory cytokines. To identify the functional links of BRAF, we used the STRING database.
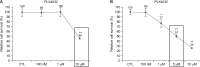
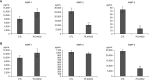
Results: Docking results illustrated PLX4032 blocked the kinase activity by exclusively binding on the serine/threonine kinase domain. STRING results indicated BRAF is functionally linked to mitogen-activated protein kinase. Both cell lines showed a dose-dependent reduction in growth rate but had a different half maximal inhibitory concentration value for PLX4032. The reaction to PLX4032 was more sensitive in the 8505C cells than in the TPC1 cells. PLX4032 induced a G2/M phase arrest in the TPC1 cells and G0/G1 in the 8505C cells. PLX4032 induced apoptosis only in the 8505C cells. With PLX4032, the TPC1 cells showed decreased levels of vascular endothelial growth factor, granulocyte-macrophage colony-stimulating factor, chemokine (C-C motif) ligand 2/monocyte chemoattractant protein 1, whereas the 8505C cells showed significantly decreased levels of IL-8, serpin E1/plasminogen activator inhibitor-1, and matrix metalloproteinase (MMP)-3.
Conclusion: PLX4032 was cytotoxic in both TPC1 and 8505C cells and induced apoptosis. In the 8505C cells, inflammatory cytokines such as IL-8 and MMP-3 were down-regulated. These findings suggest the possibility that the BRAFV600E mutation needs to target inflammatory signaling pathways in the treatment of thyroid cancer.
Keywords: BRAFV600E; Cytokines; Thyroid neoplasms; Vemurafenib.
Copyright © 2021, the Korean Surgical Society.
Conflict of interest statement
Conflict of Interest: No potential conflict of interest relevant to this article was reported.
Figures
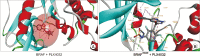
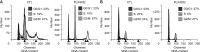

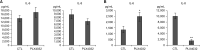

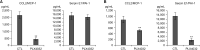
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

