Microfluidic Flow Injection Immunoassay System for Algal Toxins Determination: A Case of Study
- PMID: 33748075
- PMCID: PMC7974544
- DOI: 10.3389/fchem.2021.626630
Microfluidic Flow Injection Immunoassay System for Algal Toxins Determination: A Case of Study
Abstract
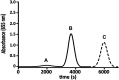
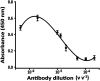
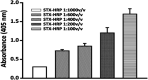
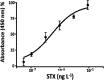
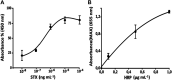
A novel flow injection microfluidic immunoassay system for continuous monitoring of saxitoxin, a lethal biotoxin, in seawater samples is presented in this article. The system consists of a preimmobilized G protein immunoaffinity column connected in line with a lab-on-chip setup. The detection of saxitoxin in seawater was carried out in two steps: an offline incubation step (competition reaction) performed between the analyte of interest (saxitoxin or Ag, as standard or seawater sample) and a tracer (an enzyme-conjugated antigen or Ag*) toward a specific polyclonal antibody. Then, the mixture was injected through a "loop" of a few μL using a six-way injection valve into a bioreactor, in line with the valve. The bioreactor consisted of a small glass column, manually filled with resin upon which G protein has been immobilized. When the mixture flowed through the bioreactor, all the antibody-antigen complex, formed during the competition step, is retained by the G protein. The tracer molecules that do not interact with the capture antibody and protein G are eluted out of the column, collected, and mixed with an enzymatic substrate directly within the microfluidic chip, via the use of two peristaltic pumps. When Ag* was present, a color change (absorbance variation, ΔAbs) of the solution is detected at a fixed wavelength (655 nm) by an optical chip docking system and registered by a computer. The amount of saxitoxin, present in the sample (or standard), that generates the variation of the intensity of the color, will be directly proportional to the concentration of the analyte in the analyzed solution. Indeed, the absorbance response increased proportionally to the enzymatic product and to the concentration of saxitoxin in the range of 3.5 × 10-7-2 × 10-5 ng ml-1 with a detection limit of 1 × 10-7 ng ml-1 (RSD% 15, S N-1 equal to 3). The immunoanalytical system has been characterized, optimized, and tested with seawater samples. This analytical approach, combined with the transportable and small-sized instrumentation, allows for easy in situ monitoring of marine water contaminations.
Keywords: FI-IA; algal toxins; immunoanalytical system; microchip flow-chamber system; saxitoxin.
Copyright © 2021 Celio, Ottaviani, Cancelliere, Di Tinno, Panjan, Sesay and Micheli.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




References
-
- Botana A. M., Otero P., Rodríguez P., Alfonso A., Botana L. M. (2013). Current situation on analysis of marine toxins. Rev. Anal. Chem. 32 (1), 15–34. 10.1515/revac-2012-0020 - DOI
-
- Campbell K., Rawn D. F., Niedzwiadek B., Elliott C. T. (2011). Paralytic shellfish poisoning (PSP) toxin binders for optical biosensor technology: problems and possibilities for the future: a review. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 28, 711–725. 10.1080/19440049.2010.531198 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

