Fabrication and Characterization of Chitosan/Poly(Lactic-Co-glycolic Acid) Core-Shell Nanoparticles by Coaxial Electrospray Technology for Dual Delivery of Natamycin and Clotrimazole
- PMID: 33748084
- PMCID: PMC7973235
- DOI: 10.3389/fbioe.2021.635485
Fabrication and Characterization of Chitosan/Poly(Lactic-Co-glycolic Acid) Core-Shell Nanoparticles by Coaxial Electrospray Technology for Dual Delivery of Natamycin and Clotrimazole
Abstract
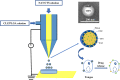
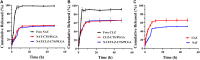
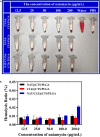
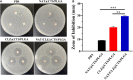
Natamycin (NAT) is the drug of choice for the treatment of fungal keratitis (FK). However, its inherent shortcomings, such as poor solubility, high dosing frequency, and long treatment cycle, need to be urgently addressed by designing a new delivery to widen its clinical utility. Growing research has confirmed that clotrimazole (CLZ) plays a significant role in fungal growth inhibition. Hence, coaxial electrospray (CO-ES) technology is used herein to prepare nano-systems with an average hydrodynamic particle size of 309-406 nm for the co-delivery of NAT and CLZ in chitosan (CTS) and poly(lactic-co-glycolic acid) (PLGA). The resulting NAT/CLZ@CTS/PLGA formulations were characterized by a transmission electron microscope (TEM) and in vitro release test. The results show that the formulations had obvious core-shell structures, uniform particle distribution, and also can sustain the release of drugs over 36 h. Furthermore, in vitro hemolysis, in vivo corneal irritation test, local allergenic test, and antifungal activity analyses are performed to evaluate the safety and efficiency of the formulations. Thus, good biosafety along with a significant anti-candidiasis effect are found in the NAT/CLZ@CTS/PLGA nanoparticles (NPs). Taken together, the results suggest that this design may provide a promising drug delivery system and a new option for the treatment of FK.
Keywords: C. albicans; clotrimazole; coaxial electrospray; core-shell structure; natamycin.
Copyright © 2021 Cui, Li, Xu, Guan, Ma, Ding and Zhang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

