Amyloid-Like Aggregation in Diseases and Biomaterials: Osmosis of Structural Information
- PMID: 33748087
- PMCID: PMC7966729
- DOI: 10.3389/fbioe.2021.641372
Amyloid-Like Aggregation in Diseases and Biomaterials: Osmosis of Structural Information
Abstract
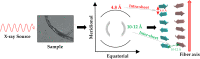
The discovery that the polypeptide chain has a remarkable and intrinsic propensity to form amyloid-like aggregates endowed with an extraordinary stability is one of the most relevant breakthroughs of the last decades in both protein/peptide chemistry and structural biology. This observation has fundamental implications, as the formation of these assemblies is systematically associated with the insurgence of severe neurodegenerative diseases. Although the ability of proteins to form aggregates rich in cross-β structure has been highlighted by recent studies of structural biology, the determination of the underlying atomic models has required immense efforts and inventiveness. Interestingly, the progressive molecular and structural characterization of these assemblies has opened new perspectives in apparently unrelated fields. Indeed, the self-assembling through the cross-β structure has been exploited to generate innovative biomaterials endowed with promising mechanical and spectroscopic properties. Therefore, this structural motif has become the fil rouge connecting these diversified research areas. In the present review, we report a chronological recapitulation, also performing a survey of the structural content of the Protein Data Bank, of the milestones achieved over the years in the characterization of cross-β assemblies involved in the insurgence of neurodegenerative diseases. A particular emphasis is given to the very recent successful elucidation of amyloid-like aggregates characterized by remarkable molecular and structural complexities. We also review the state of the art of the structural characterization of cross-β based biomaterials by highlighting the benefits of the osmosis of information between these two research areas. Finally, we underline the new promising perspectives that recent successful characterizations of disease-related amyloid-like assemblies can open in the biomaterial field.
Keywords: amino acid aggregation; amyloid aggregates; biomaterials; cross-β structure; glutamine rich structures; peptide-based hydrogels.
Copyright © 2021 Balasco, Diaferia, Morelli, Vitagliano and Accardo.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



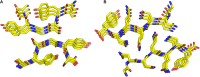

References
-
- Adams D. J., Mullen L. M., Berta M., Chen L., Frith W. J. (2010). Relationship between molecular structure, gelation behaviour and gel properties of Fmoc-dipeptides. Soft Matter 6 1971–1980. 10.1039/B921863G - DOI
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

