Islet sympathetic innervation and islet neuropathology in patients with type 1 diabetes
- PMID: 33753784
- PMCID: PMC7985489
- DOI: 10.1038/s41598-021-85659-8
Islet sympathetic innervation and islet neuropathology in patients with type 1 diabetes
Abstract
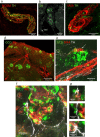
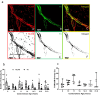
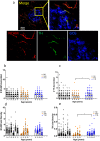
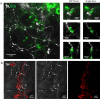
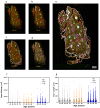
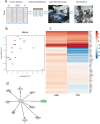
Dysregulation of glucagon secretion in type 1 diabetes (T1D) involves hypersecretion during postprandial states, but insufficient secretion during hypoglycemia. The sympathetic nervous system regulates glucagon secretion. To investigate islet sympathetic innervation in T1D, sympathetic tyrosine hydroxylase (TH) axons were analyzed in control non-diabetic organ donors, non-diabetic islet autoantibody-positive individuals (AAb), and age-matched persons with T1D. Islet TH axon numbers and density were significantly decreased in AAb compared to T1D with no significant differences observed in exocrine TH axon volume or lengths between groups. TH axons were in close approximation to islet α-cells in T1D individuals with long-standing diabetes. Islet RNA-sequencing and qRT-PCR analyses identified significant alterations in noradrenalin degradation, α-adrenergic signaling, cardiac β-adrenergic signaling, catecholamine biosynthesis, and additional neuropathology pathways. The close approximation of TH axons at islet α-cells supports a model for sympathetic efferent neurons directly regulating glucagon secretion. Sympathetic islet innervation and intrinsic adrenergic signaling pathways could be novel targets for improving glucagon secretion in T1D.
Conflict of interest statement
The authors declare no competing interests.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

