Striatal transcriptome changes linked to drug-induced repetitive behaviors
- PMID: 33759265
- PMCID: PMC8330602
- DOI: 10.1111/ejn.15116
Striatal transcriptome changes linked to drug-induced repetitive behaviors
Abstract
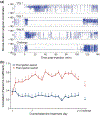
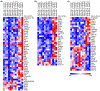
Disruptive or excessive repetitive motor patterns (stereotypies) are cardinal symptoms in numerous neuropsychiatric disorders. Stereotypies are also evoked by psychomotor stimulants such as amphetamine. The acquisition of motor sequences is paralleled by changes in activity patterns in the striatum, and stereotypies have been linked to abnormal plasticity in these reinforcement-related circuits. Here, we designed experiments in mice to identify transcriptomic changes that underlie striatal plasticity occurring alongside the development of drug-induced stereotypic behavior. We identified three schedules of amphetamine treatment inducing different degrees of stereotypy and used bulk RNAseq to compare striatal gene expression changes among groups of mice treated with the different drug-dose schedules and vehicle-treated, cage-mate controls. Mice were identified as naïve, sensitized, or tolerant to drug-induced stereotypy. All drug-treated groups exhibited expression changes in genes that encode members of the extracellular signal-regulated kinase (ERK) cascades known to regulate psychomotor stimulant responses. In the sensitized group with the most prolonged stereotypy, we found dysregulation of 20 genes that were not changed in other groups. Gene set enrichment analysis indicated highly significant overlap with genes regulated by neuregulin 1 (Nrg1). Nrg1 is known to be a schizophrenia and autism susceptibility gene that encodes a ligand for Erb-B receptors, which are involved in neuronal migration, myelination, and cell survival, including that of dopamine-containing neurons. Stimulant abuse is a risk factor for schizophrenia onset, and these two disorders share behavioral stereotypy phenotypes. Our results raise the possibility that drug-induced sensitization of the Nrg1 signaling pathway might underlie these links.
Keywords: amphetamine; restricted repetitive behavior; sensitization; stereotypy; tolerance.
© 2021 Federation of European Neuroscience Societies and John Wiley & Sons Ltd.
Conflict of interest statement
CONFLICT OF INTEREST
The authors report no competing interests.
Figures

Similar articles
-
Severe drug-induced repetitive behaviors and striatal overexpression of VAChT in ChAT-ChR2-EYFP BAC transgenic mice.Front Neural Circuits. 2014 May 28;8:57. doi: 10.3389/fncir.2014.00057. eCollection 2014. Front Neural Circuits. 2014. PMID: 24904300 Free PMC article.
-
Responses of neurons in dorsal striatum during amphetamine-induced focused stereotypy.Psychopharmacology (Berl). 1997 Apr;130(4):343-51. doi: 10.1007/s002130050249. Psychopharmacology (Berl). 1997. PMID: 9160850
-
The role of D1 and D2 dopamine receptors in oral stereotypy induced by dopaminergic stimulation of the ventrolateral striatum.Neuroscience. 1990;39(1):59-67. doi: 10.1016/0306-4522(90)90221-o. Neuroscience. 1990. PMID: 1982467
-
Amphetamine stereotypy, the basal ganglia, and the "selection problem".Behav Brain Res. 2012 Jun 1;231(2):297-308. doi: 10.1016/j.bbr.2011.11.003. Epub 2011 Nov 11. Behav Brain Res. 2012. PMID: 22101067 Review.
-
Enduring changes in brain and behavior produced by chronic amphetamine administration: a review and evaluation of animal models of amphetamine psychosis.Brain Res. 1986 Jun;396(2):157-98. doi: 10.1016/s0006-8993(86)80193-7. Brain Res. 1986. PMID: 3527341 Review.
Cited by
-
Genetic labeling reveals spatial and cellular expression pattern of neuregulin 1 in mouse brain.Cell Biosci. 2023 May 5;13(1):79. doi: 10.1186/s13578-023-01032-4. Cell Biosci. 2023. PMID: 37147705 Free PMC article.
-
Cannabinoid Receptor 1 Is Required for Neurodevelopment of Striosome-Dendron Bouquets.eNeuro. 2022 Apr 11;9(2):ENEURO.0318-21.2022. doi: 10.1523/ENEURO.0318-21.2022. Print 2022 Mar-Apr. eNeuro. 2022. PMID: 35361667 Free PMC article.
-
Fluoxetine potentiates methylphenidate-induced behavioral responses: Enhanced locomotion or stereotypies and facilitated acquisition of cocaine self-administration.Addict Neurosci. 2023 Dec 15;9:100131. doi: 10.1016/j.addicn.2023.100131. Epub 2023 Sep 26. Addict Neurosci. 2023. PMID: 38222942 Free PMC article.
-
A transient protein folding response targets aggregation in the early phase of TDP-43-mediated neurodegeneration.Nat Commun. 2024 Feb 19;15(1):1508. doi: 10.1038/s41467-024-45646-9. Nat Commun. 2024. PMID: 38374041 Free PMC article.
-
Quantitative trait locus analysis for endophenotypes reveals genetic substrates of core symptom domains and neurocognitive function in autism spectrum disorder.Transl Psychiatry. 2022 Sep 24;12(1):407. doi: 10.1038/s41398-022-02179-3. Transl Psychiatry. 2022. PMID: 36153334 Free PMC article.
References
-
- Abe Y, Namba H, Zheng Y & Nawa H (2009) In situ hybridization reveals developmental regulation of ErbB1-4 mRNA expression in mouse midbrain: implication of ErbB receptors for dopaminergic neurons. Neuroscience, 161, 95–110. - PubMed
-
- American Psychiatric Association, D.-V. (ed) (2013) American Psychiatric Association: Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition., Washington, DC.
-
- Angrist B, Sathananthan G, Wilk S & Gershon S (1974) Amphetamine psychosis: behavioral and biochemical aspects. Journal of Psychiatric Research, 11, 13–23. - PubMed
-
- Barnes TD, Kubota Y, Hu D, Jin DZ & Graybiel AM (2005) Activity of striatal neurons reflects dynamic encoding and recoding of procedural memories. Nature, 437, 1158–1161. - PubMed
-
- Berke JD & Hyman SE (2000) Addiction, dopamine, and the molecular mechanisms of memory. Neuron, 25, 515–532. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

