Cytisine versus varenicline for smoking cessation in New Zealand indigenous Māori: a randomized controlled trial
- PMID: 33761149
- PMCID: PMC8519028
- DOI: 10.1111/add.15489
Cytisine versus varenicline for smoking cessation in New Zealand indigenous Māori: a randomized controlled trial
Abstract
Aim: To determine whether cytisine was at least as effective as varenicline in supporting smoking abstinence for ≥ 6 months in New Zealand indigenous Māori or whānau (extended-family) of Māori, given the high smoking prevalence in this population.
Design: Pragmatic, open-label, randomized, community-based non-inferiority trial.
Setting: Bay of Plenty, Tokoroa and Lakes District Health Board regions of New Zealand.
Participants: Adult daily smokers who identified as Māori or whānau of Māori, were motivated to quit in the next 2 weeks, were aged ≥ 18 years and were eligible for subsidized varenicline. Recruitment used multi-media advertising.
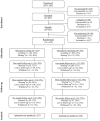
Interventions: A total of 679 people were randomly assigned (1 : 1) to receive a prescription for 12 weeks of cytisine or varenicline, plus low-intensity cessation behavioural support from the prescribing doctor and community stop-smoking services or a research assistant. Day 5 of treatment was the designated quit date.
Measurements: The primary outcome was carbon monoxide-verified continuous abstinence at 6 months, analysed as intention-to-treat (with multiple imputation for missing data). Secondary outcomes measured at 1, 3, 6 and 12 months post-quit date included: self-reported continuous abstinence, 7-day point prevalence abstinence, cigarettes per day, time to (re)lapse, adverse events, treatment adherence/compliance and acceptability, nicotine withdrawal/urge to smoke and health-care utilization/health-related quality of life.
Findings: Verified continuous abstinence rates at 6 months post-quit date were 12.1% (41 of 337) for cytisine versus 7.9% (27 of 342) for varenicline [risk difference 4.29%, 95% confidence interval (CI) = -0.22 to 8.79; relative risk 1.55; 95% CI = 0.97-2.46]. Sensitivity analyses confirmed that the findings were robust. Self-reported adverse events over 6 months occurred significantly more frequently in the varenicline group (cytisine: 313 events in 111 participants; varenicline: 509 events in 138 participants, incidence rate ratio 0.56, 95% CI = 0.49-0.65, P < 0.001) compared with the cytisine group. Common adverse events were headache, nausea and difficulty sleeping.
Conclusion: A randomized controlled trial found that cytisine was at least as effective as varenicline at supporting smoking abstinence in New Zealand indigenous Māori or whānau (extended-family) of Māori, with significantly fewer adverse events.
Trial registration: ClinicalTrials.gov NCT02957786.
Keywords: Cytisine; indigenous; non-inferiority; smoking; trial; varenicline.
© 2021 The Authors. Addiction published by John Wiley & Sons Ltd on behalf of Society for the Study of Addiction.
Figures
References
-
- Tutka P., Vinnikov D., Courtney R. J., Benowitz N. L. Cytisine for nicotine addiction treatment: a review of pharmacology, therapeutics and an update of clinical trial evidence for smoking cessation. Addiction 2019; 114: 1951–1969. - PubMed
-
- Walker N., Bullen C., Barnes J., McRobbie H., Tutka P., Raw M., et al. Getting cytisine licensed for use worldwide: a call to action. [Editorial]. Addiction 2016; 111: 1895–1898. - PubMed
-
- Coe J. W., Brooks P. R., Vetelino M. G., Wirtz M. C., Arnold E. P., Huang J., et al. Varenicline: an alpha4beta2 nicotinic receptor partial agonist for smoking cessation. J Med Chem 2005; 48: 3474–3477. - PubMed
-
- Jeong S.‐H., Newcombe D., Sheridan J., Tingle M. Pharmacokinetics of cytisine, an α4β2 nicotinic receptor partial agonist, in healthy smokers following a single dose. Drug Test Anal 2015; 7: 475–482. - PubMed
-
- Obach R., Reed‐Hagen A., Krueger S., Obach B., O'Connell T., Zandi K., et al. Metabolism and disposition of varenicline, a selective alpha4beta2 acetylcholine receptor partial agonist, in vivo and in vitro . Drug Metab Dispos 2006; 34: 121–130. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

