Tackling tumor microenvironment through epigenetic tools to improve cancer immunotherapy
- PMID: 33761971
- PMCID: PMC7992805
- DOI: 10.1186/s13148-021-01046-0
Tackling tumor microenvironment through epigenetic tools to improve cancer immunotherapy
Abstract
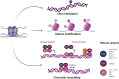
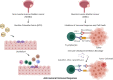
Background: Epigenetic alterations are known contributors to cancer development and aggressiveness. Additional to alterations in cancer cells, aberrant epigenetic marks are present in cells of the tumor microenvironment, including lymphocytes and tumor-associated macrophages, which are often overlooked but known to be a contributing factor to a favorable environment for tumor growth. Therefore, the main aim of this review is to give an overview of the epigenetic alterations affecting immune cells in the tumor microenvironment to provoke an immunosuppressive function and contribute to cancer development. Moreover, immunotherapy is briefly discussed in the context of epigenetics, describing both its combination with epigenetic drugs and the need for epigenetic biomarkers to predict response to immune checkpoint blockage.
Main body: Combining both topics, epigenetic machinery plays a central role in generating an immunosuppressive environment for cancer growth, which creates a barrier for immunotherapy to be successful. Furthermore, epigenetic-directed compounds may not only affect cancer cells but also immune cells in the tumor microenvironment, which could be beneficial for the clinical response to immunotherapy.
Conclusion: Thus, modulating epigenetics in combination with immunotherapy might be a promising therapeutic option to improve the success of this therapy. Further studies are necessary to (1) understand in depth the impact of the epigenetic machinery in the tumor microenvironment; (2) how the epigenetic machinery can be modulated according to tumor type to increase response to immunotherapy and (3) find reliable biomarkers for a better selection of patients eligible to immunotherapy.
Keywords: Bladder cancer; Epigenetics; Immunotherapy; Therapy; Tumor microenvironment.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

Similar articles
-
Epigenetics and immunotherapy: The current state of play.Mol Immunol. 2017 Jul;87:227-239. doi: 10.1016/j.molimm.2017.04.012. Epub 2017 May 14. Mol Immunol. 2017. PMID: 28511092 Review.
-
Leveraging epigenetics to enhance the efficacy of immunotherapy.Clin Epigenetics. 2021 May 17;13(1):115. doi: 10.1186/s13148-021-01100-x. Clin Epigenetics. 2021. PMID: 34001289 Free PMC article. Review.
-
The emerging role of epigenetic therapeutics in immuno-oncology.Nat Rev Clin Oncol. 2020 Feb;17(2):75-90. doi: 10.1038/s41571-019-0266-5. Epub 2019 Sep 23. Nat Rev Clin Oncol. 2020. PMID: 31548600 Free PMC article. Review.
-
Epigenetic modulation of antitumor immunity for improved cancer immunotherapy.Mol Cancer. 2021 Dec 20;20(1):171. doi: 10.1186/s12943-021-01464-x. Mol Cancer. 2021. PMID: 34930302 Free PMC article. Review.
-
Tumor microenvironment noise-induced polarization: the main challenge in macrophages' immunotherapy for cancer.Mol Cell Biochem. 2025 Jun;480(6):3735-3747. doi: 10.1007/s11010-025-05205-2. Epub 2025 Jan 19. Mol Cell Biochem. 2025. PMID: 39827422 Free PMC article. Review.
Cited by
-
Molecular Crosstalk between Chromatin Remodeling and Tumor Microenvironment in Multiple Myeloma.Curr Oncol. 2022 Dec 5;29(12):9535-9549. doi: 10.3390/curroncol29120749. Curr Oncol. 2022. PMID: 36547163 Free PMC article. Review.
-
An Immune-Enhancing Injectable Hydrogel Loaded with Esketamine and DDP Promotes Painless Immunochemotherapy to Inhibit Breast Cancer Growth.Adv Healthc Mater. 2024 Nov;13(29):e2401373. doi: 10.1002/adhm.202401373. Epub 2024 Aug 9. Adv Healthc Mater. 2024. PMID: 39118566 Free PMC article.
-
Epigenetic Regulation of Stromal and Immune Cells and Therapeutic Targets in the Tumor Microenvironment.Biomolecules. 2025 Jan 6;15(1):71. doi: 10.3390/biom15010071. Biomolecules. 2025. PMID: 39858465 Free PMC article. Review.
-
Molecular basis of epigenetic regulation in cancer diagnosis and treatment.Front Genet. 2022 Aug 24;13:885635. doi: 10.3389/fgene.2022.885635. eCollection 2022. Front Genet. 2022. PMID: 36092905 Free PMC article. Review.
-
Epigenetics of Dendritic Cells in Tumor Immunology.Cancers (Basel). 2022 Feb 24;14(5):1179. doi: 10.3390/cancers14051179. Cancers (Basel). 2022. PMID: 35267487 Free PMC article. Review.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

