A Dengue Virus Serotype 1 mRNA-LNP Vaccine Elicits Protective Immune Responses
- PMID: 33762420
- PMCID: PMC8315947
- DOI: 10.1128/JVI.02482-20
A Dengue Virus Serotype 1 mRNA-LNP Vaccine Elicits Protective Immune Responses
Abstract
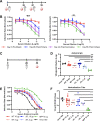
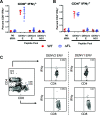
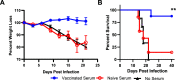
Dengue virus (DENV) is the most common vector-borne viral disease, with nearly 400 million worldwide infections each year concentrated in the tropical and subtropical regions of the world. Severe dengue complications are often associated with a secondary heterotypic infection of one of the four circulating serotypes. In this scenario, humoral immune responses targeting cross-reactive, poorly neutralizing epitopes can lead to increased infectivity of susceptible cells via antibody-dependent enhancement (ADE). In this way, antibodies produced in response to infection or vaccination are capable of contributing to enhanced disease in subsequent infections. Currently, there are no available therapeutics to combat DENV disease, and there is an urgent need for a safe and efficacious vaccine. Here, we developed a nucleotide-modified mRNA vaccine encoding the membrane and envelope structural proteins from DENV serotype 1 encapsulated in lipid nanoparticles (prM/E mRNA-LNP). Vaccination of mice elicited robust antiviral immune responses comparable to viral infection, with high levels of neutralizing antibody titers and antiviral CD4+ and CD8+ T cells. Immunocompromised AG129 mice vaccinated with the prM/E mRNA-LNP vaccine were protected from a lethal DENV challenge. Vaccination with either a wild-type vaccine or a vaccine with mutations in the immunodominant fusion loop epitope elicited equivalent humoral and cell-mediated immune responses. Neutralizing antibodies elicited by the vaccine were sufficient to protect against a lethal challenge. Both vaccine constructs demonstrated serotype-specific immunity with minimal serum cross-reactivity and reduced ADE in comparison to a live DENV1 viral infection.IMPORTANCE With 400 million worldwide infections each year, dengue is the most common vector-borne viral disease. Forty percent of the world's population is at risk, with dengue experiencing consistent geographic spread over the years. With no therapeutics available and vaccines performing suboptimally, the need for an effective dengue vaccine is urgent. Here, we develop and characterize a novel mRNA vaccine encoding the dengue serotype 1 envelope and premembrane structural proteins that is delivered via a lipid nanoparticle. Our DENV1 prM/E mRNA-LNP vaccine induces neutralizing antibody and cellular immune responses in immunocompetent mice and protects an immunocompromised mouse from a lethal DENV challenge. Existing antibodies against dengue can enhance subsequent infections via antibody-dependent enhancement (ADE). Importantly our vaccine induced only serotype-specific immune responses and did not induce ADE.
Keywords: dengue fever; mRNA vaccine; vaccines.
Copyright © 2021 American Society for Microbiology.
Figures




References
-
- Bhatt S, Gething PW, Brady OJ, Messina JP, Farlow AW, Moyes CL, Drake JM, Brownstein JS, Hoen AG, Sankoh O, Myers MF, George DB, Jaenisch T, Wint GRW, Simmons CP, Scott TW, Farrar JJ, Hay SI. 2013. The global distribution and burden of dengue. Nature 496:504–507. 10.1038/nature12060. - DOI - PMC - PubMed
-
- World Health Organization. 2020. Dengue and severe dengue. World Health Organization, Geneva, Switzerland. https://www.who.int/news-room/fact-sheets/detail/dengue-and-severe-dengue. Accessed 5 August 2020.
-
- World Health Organization. 2009. Dengue: guidelines for diagnosis, treatment, prevention, and control, new ed. World Health Organization, Geneva. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

