SVEP1 is a human coronary artery disease locus that promotes atherosclerosis
- PMID: 33762433
- PMCID: PMC8109261
- DOI: 10.1126/scitranslmed.abe0357
SVEP1 is a human coronary artery disease locus that promotes atherosclerosis
Abstract
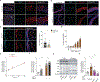
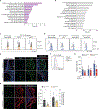
A low-frequency variant of sushi, von Willebrand factor type A, EGF, and pentraxin domain-containing protein 1 (SVEP1), an extracellular matrix protein, is associated with risk of coronary disease in humans independent of plasma lipids. Despite a robust statistical association, if and how SVEP1 might contribute to atherosclerosis remained unclear. Here, using Mendelian randomization and complementary mouse models, we provide evidence that SVEP1 promotes atherosclerosis in humans and mice and is expressed by vascular smooth muscle cells (VSMCs) within the atherosclerotic plaque. VSMCs also interact with SVEP1, causing proliferation and dysregulation of key differentiation pathways, including integrin and Notch signaling. Fibroblast growth factor receptor transcription increases in VSMCs interacting with SVEP1 and is further increased by the coronary disease-associated SVEP1 variant p.D2702G. These effects ultimately drive inflammation and promote atherosclerosis. Together, our results suggest that VSMC-derived SVEP1 is a proatherogenic factor and support the concept that pharmacological inhibition of SVEP1 should protect against atherosclerosis in humans.
Copyright © 2021 The Authors, some rights reserved; exclusive licensee American Association for the Advancement of Science. No claim to original U.S. Government Works.
Conflict of interest statement
Figures





References
-
- Weber C, Noels H, Atherosclerosis: current pathogenesis and therapeutic options. Nat Med 17, 1410–1422 (2011). - PubMed
-
- Dzau VJ, Braun-Dullaeus RC, Sedding DG, Vascular proliferation and atherosclerosis: new perspectives and therapeutic strategies. Nat Med 8, 1249–1256 (2002). - PubMed
-
- Liu X, Ntambi JM, Atherosclerosis: keep your macrophages in shape. Nat Med 15, 1357–1358 (2009). - PubMed
-
- Hansson GK, Klareskog L, Pulling down the plug on atherosclerosis: cooling down the inflammasome. Nat Med 17, 790–791 (2011). - PubMed
-
- Ross R, Genetically modified mice as models of transplant atherosclerosis. Nat Med 2, 527–528 (1996). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 HL125838/HL/NHLBI NIH HHS/United States
- T32 HL134635/HL/NHLBI NIH HHS/United States
- R01 HL159171/HL/NHLBI NIH HHS/United States
- T32 HL007081/HL/NHLBI NIH HHS/United States
- T32 GM007200/GM/NIGMS NIH HHS/United States
- UM1 HG008853/HG/NHGRI NIH HHS/United States
- P30 CA091842/CA/NCI NIH HHS/United States
- R01 HL139714/HL/NHLBI NIH HHS/United States
- R01 HL131961/HL/NHLBI NIH HHS/United States
- R01 HL053325/HL/NHLBI NIH HHS/United States
- R01 HL138466/HL/NHLBI NIH HHS/United States
- P30 DK020579/DK/NIDDK NIH HHS/United States
- UL1 TR002345/TR/NCATS NIH HHS/United States
- I01 BX003415/BX/BLRD VA/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

