The effect of an orally-dosed Caralluma Fimbriata extract on appetite control and body composition in overweight adults
- PMID: 33762661
- PMCID: PMC7991653
- DOI: 10.1038/s41598-021-86108-2
The effect of an orally-dosed Caralluma Fimbriata extract on appetite control and body composition in overweight adults
Abstract
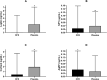
To examine the effect of a Caralluma Fimbriata extract (CFE) on biomarkers of satiety and body composition in overweight adults. A double-blind, randomised, placebo controlled trial to examine the effect of a Caralluma Fimbriata extract (CFE) on biomarkers of satiety and body composition in overweight adults. Eighty-three men and women aged between 20 and 50 years of age completed 16 weeks of daily supplementation with either CFE or placebo. Plasma cardiometabolic (lipid profile, glucose, insulin) and satiety (ghrelin, leptin, neuropeptideY) biomarkers, body composition, diet history and gastrointenstinal function were assessed at baseline, weeks 4, 8, 12 and 16. Subjects in the CFE and placebo groups were well matched and predominatly female 93% and 87.5%, with a mean age of 40.9 ± 6.7 and 39.5 ± 7.5 years and body mass index (BMI) of 30.0 ± 3.1 and 30.2 ± 2.9 kg/m2 respectively. There was a significant difference in plasma leptin concentration change between groups at week 16 (p = 0.04), with the placebo group increasing concentration (2.27 ± 4.80 ng/mL) while the CFE group (0.05 ± 4.69 ng/mL) remained the same. At week 16, the CFE group had significantly reduced their calorie intake from baseline compared to the placebo group (245 cal vs 15.8 cal respectively p < 0.01). The CFE group also had a significant reduction in waist circumference of 2.7 cm compared to an increase of 0.3 cm in the placebo group (p = 0.02). A weight increase from baseline was seen in the placebo group that was not observed in the CFE group (1.33 kg weight gain vs 0.37 kg weight loss respectively; p = 0.03). The placebo group also had a significant increase in fat mass, android fat mass, BMI and leptin compared to the CFE group (p = 0.04, 0.02, < 0.01 respectively). CFE was effective at maintaining bodyweight during a non-calorie controlled diet compared to a placebo. The mechanism responsible for this action is requiring further research and could be due to an increase in satiety receptor sensitivity.
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
A pilot study investigating the effect of Caralluma fimbriata extract on the risk factors of metabolic syndrome in overweight and obese subjects: a randomised controlled clinical trial.Complement Ther Med. 2013 Jun;21(3):180-9. doi: 10.1016/j.ctim.2013.01.004. Epub 2013 Feb 23. Complement Ther Med. 2013. PMID: 23642949 Clinical Trial.
-
Phytochemical analysis and effects on ingestive behaviour of a Caralluma fimbriata extract.Food Chem Toxicol. 2017 Oct;108(Pt A):63-73. doi: 10.1016/j.fct.2017.07.027. Epub 2017 Jul 13. Food Chem Toxicol. 2017. PMID: 28713048
-
Effect of Caralluma fimbriata extract on appetite, food intake and anthropometry in adult Indian men and women.Appetite. 2007 May;48(3):338-44. doi: 10.1016/j.appet.2006.09.013. Epub 2006 Nov 13. Appetite. 2007. PMID: 17097761 Clinical Trial.
-
The use of Caralluma fimbriata as an appetite suppressant and weight loss supplement: a systematic review and meta-analysis of clinical trials.BMC Complement Med Ther. 2021 Nov 10;21(1):279. doi: 10.1186/s12906-021-03450-8. BMC Complement Med Ther. 2021. PMID: 34758791 Free PMC article.
-
Efficacy of Olibra: a 12-week randomized controlled trial and a review of earlier studies.J Diabetes Sci Technol. 2012 May 1;6(3):695-708. doi: 10.1177/193229681200600326. J Diabetes Sci Technol. 2012. PMID: 22768902 Free PMC article. Review.
Cited by
-
Caralluma fimbriata Extract Improves Vascular Dysfunction in Obese Mice Fed a High-Fat Diet.Nutrients. 2024 Dec 12;16(24):4296. doi: 10.3390/nu16244296. Nutrients. 2024. PMID: 39770917 Free PMC article.
-
Delving the Role of Caralluma fimbriata: An Edible Wild Plant to Mitigate the Biomarkers of Metabolic Syndrome.Oxid Med Cell Longev. 2022 Jun 20;2022:5720372. doi: 10.1155/2022/5720372. eCollection 2022. Oxid Med Cell Longev. 2022. PMID: 35770046 Free PMC article. Review.
-
Efficacy and safety of Obex® in overweight and obese subjects: a randomised, double-blind, placebo-controlled clinical trial.BMC Complement Med Ther. 2023 Feb 20;23(1):58. doi: 10.1186/s12906-023-03847-7. BMC Complement Med Ther. 2023. PMID: 36804035 Free PMC article. Clinical Trial.
-
Role of pancreatic lipase inhibition in obesity treatment: mechanisms and challenges towards current insights and future directions.Int J Obes (Lond). 2025 Mar;49(3):492-506. doi: 10.1038/s41366-025-01729-1. Epub 2025 Feb 27. Int J Obes (Lond). 2025. PMID: 40016558 Free PMC article. Review.
-
Medicinal Plant Extracts against Cardiometabolic Risk Factors Associated with Obesity: Molecular Mechanisms and Therapeutic Targets.Pharmaceuticals (Basel). 2024 Jul 21;17(7):967. doi: 10.3390/ph17070967. Pharmaceuticals (Basel). 2024. PMID: 39065815 Free PMC article. Review.
References
-
- World Health Organisation. 2019 [cited 2019 30/09/2019]; Obesity and Overweight]. Available from: https://www.who.int/westernpacific/health-topics/obesity-and-overweight.
-
- The Obesity Collective, Weighing In: Australia’s Growing Obesity Epidemic. 2019, The Obesity Collective.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

