[18F]F13640, a 5-HT1A Receptor Radiopharmaceutical Sensitive to Brain Serotonin Fluctuations
- PMID: 33762906
- PMCID: PMC7982540
- DOI: 10.3389/fnins.2021.622423
[18F]F13640, a 5-HT1A Receptor Radiopharmaceutical Sensitive to Brain Serotonin Fluctuations
Abstract
Introduction: Serotonin is involved in a variety of physiological functions and brain disorders. In this context, efforts have been made to investigate the in vivo fluctuations of this neurotransmitter using positron emission tomography (PET) imaging paradigms. Since serotonin is a full agonist, it binds preferentially to G-protein coupled receptors. In contrast, antagonist PET ligands additionally interact with uncoupled receptors. This could explain the lack of sensitivity to serotonin fluctuations of current 5-HT1A radiopharmaceuticals which are mainly antagonists and suggests that agonist radiotracers would be more appropriate to measure changes in neurotransmitter release. The present study evaluated the sensitivity to endogenous serotonin release of a recently developed, selective 5-HT1A receptor PET radiopharmaceutical, the agonist [18F]F13640 (a.k.a. befiradol or NLX-112).
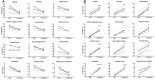
Materials and methods: Four cats each underwent three PET scans with [18F]F13640, i.e., a control PET scan of 90 min, a PET scan preceded 30 min before by an intravenous injection 1 mg/kg of d-fenfluramine, a serotonin releaser (blocking challenge), and a PET scan comprising the intravenous injection of 1 mg/kg of d-fenfluramine 30 min after the radiotracer injection (displacement challenge). Data were analyzed with regions of interest and voxel-based approaches. A lp-ntPET model approach was implemented to determine the dynamic of serotonin release during the challenge study.
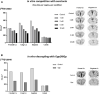
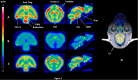
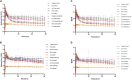
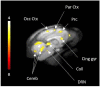
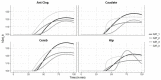
Results: D-fenfluramine pretreatment elicited a massive inhibition of [18F]F13640 labeling in regions known to express 5-HT1A receptors, e.g., raphe nuclei, hippocampus, thalamus, anterior cingulate cortex, caudate putamen, occipital, frontal and parietal cortices, and gray matter of cerebellum. Administration of d-fenfluramine during PET acquisition indicates changes in occupancy from 10% (thalamus) to 31% (gray matter of cerebellum) even though the dissociation rate of [18F]F13640 over the 90 min acquisition time was modest. The lp-ntPET simulation succeeded in differentiating the control and challenge conditions.
Conclusion: The present findings demonstrate that labeling of 5-HT1A receptors with [18F]F13640 is sensitive to serotonin concentration fluctuations in vivo. Although the data underline the need to perform longer PET scan to ensure accurate measure of displacement, they support clinical development of [18F]F13640 as a tool to explore experimental paradigms involving physiological or pathological (neurological or neuropsychiatric pathologies) fluctuations of extracellular serotonin.
Keywords: 5-HT1A receptors; PET imaging; [18F]F13640; agonist; fenfluramine; serotonin release.
Copyright © 2021 Colom, Vidal, Fieux, Redoute, Costes, Lavenne, Mérida, Irace, Iecker, Bouillot, Billard, Newman-Tancredi and Zimmer.
Conflict of interest statement
AN-T is employed by Neurolixis. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

