Antifibrotic effects of eupatilin on TGF-β1-treated human vocal fold fibroblasts
- PMID: 33765087
- PMCID: PMC7993872
- DOI: 10.1371/journal.pone.0249041
Antifibrotic effects of eupatilin on TGF-β1-treated human vocal fold fibroblasts
Abstract
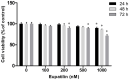
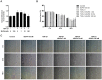
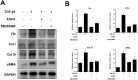
Vocal fold scarring is a major cause of dysphonia. Vocal fold fibroblasts (VFFs) and the TGF-β signaling pathway play important roles in scar formation. Eupatilin, a chromone derivative of the Artemisia species, is a traditional folk remedy for wound healing. However, until recently, few studies investigated the therapeutic effects of eupatilin. We investigated the antifibrogenic effects of eupatilin on TGF-β1-treated human vocal fold fibroblasts (hVFFs). The optimal concentration of eupatilin was determined by a cell viability assay. Western blotting was used to measure the expression of alpha-smooth muscle actin during myofibroblast differentiation, fibronectin (FN), collagen type I (Col I), and collagen type III (Col III) extracellular matrix proteins, and Smad2, Smad3, and p38 in the fibrotic pathway. Measurements were made before and after eupatilin treatment. Eupatilin at 100 nM was shown to be safe for use in hVFFs. TGF-β1 induced hVFFs to proliferate and differentiate into myofibroblasts and increased Col III and FN synthesis in a time- and dose-dependent manner. Eupatilin suppressed TGF-β1-induced hVFF proliferation and differentiation into myofibroblasts through the Smad and p38 signaling pathways. Furthermore, eupatilin inhibited TGF-β1-induced FN, Col I, and Col III synthesis in hVFFs. Our in vitro findings show that eupatilin effectively suppressed TGF-β1-induced fibrotic changes in hVFFs via the Smad and p38 signaling pathways. Thus, eupatilin may be considered a novel therapeutic agent for the treatment of vocal fold fibrosis.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


Similar articles
-
Cryotherapy has antifibrotic and regenerative effects on human vocal fold fibroblasts.Laryngoscope. 2019 Apr;129(4):E143-E150. doi: 10.1002/lary.27499. Epub 2018 Oct 12. Laryngoscope. 2019. PMID: 30315572
-
The Antiproliferative and Antifibrotic Effects of Cisplatin on Primary Human Vocal Fold Fibroblasts.ORL J Otorhinolaryngol Relat Spec. 2020;82(4):188-200. doi: 10.1159/000506708. Epub 2020 Apr 8. ORL J Otorhinolaryngol Relat Spec. 2020. PMID: 32268330
-
Photodynamic therapy induces antifibrotic alterations in primary human vocal fold fibroblasts.Laryngoscope. 2018 Sep;128(9):E323-E331. doi: 10.1002/lary.27219. Epub 2018 Apr 18. Laryngoscope. 2018. PMID: 29668038
-
Corneal fibroblast collagen type IV negative feedback modulation of TGF beta: A fibrosis modulating system likely active in other organs.Matrix Biol. 2022 May;109:162-172. doi: 10.1016/j.matbio.2022.04.002. Epub 2022 Apr 11. Matrix Biol. 2022. PMID: 35421526 Review.
-
Naringenin: A Promising Therapeutic Agent against Organ Fibrosis.Oxid Med Cell Longev. 2021 Nov 11;2021:1210675. doi: 10.1155/2021/1210675. eCollection 2021. Oxid Med Cell Longev. 2021. PMID: 34804359 Free PMC article. Review.
Cited by
-
The mechanism of microglia-mediated immune inflammation in ischemic stroke and the role of natural botanical components in regulating microglia: A review.Front Immunol. 2023 Feb 2;13:1047550. doi: 10.3389/fimmu.2022.1047550. eCollection 2022. Front Immunol. 2023. PMID: 36818470 Free PMC article. Review.
-
Eupatilin unveiled: An in-depth exploration of research advancements and clinical therapeutic prospects.J Transl Int Med. 2025 Apr 24;13(2):104-117. doi: 10.1515/jtim-2025-0016. eCollection 2025 Apr. J Transl Int Med. 2025. PMID: 40443403 Free PMC article.
-
[Therapeutic mechanism of Shenbing Decoction Ⅲ for renal fibrosis in chronic kidney disease: a study with network pharmacology, molecular docking and validation in rats].Nan Fang Yi Ke Da Xue Xue Bao. 2023 Jun 20;43(6):924-934. doi: 10.12122/j.issn.1673-4254.2023.06.07. Nan Fang Yi Ke Da Xue Xue Bao. 2023. PMID: 37439164 Free PMC article. Chinese.
-
Eupatilin mitigates Gestational diabetes in streptozotocin-induced diabetic pregnant rats through the Regulation of inflammation and oxidative stress.Heliyon. 2024 May 8;10(10):e30911. doi: 10.1016/j.heliyon.2024.e30911. eCollection 2024 May 30. Heliyon. 2024. PMID: 38818188 Free PMC article.
-
Eupatilin Ameliorates Hepatic Fibrosis and Hepatic Stellate Cell Activation by Suppressing β-catenin/PAI-1 Pathway.Int J Mol Sci. 2023 Mar 21;24(6):5933. doi: 10.3390/ijms24065933. Int J Mol Sci. 2023. PMID: 36983006 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

