Increased LRRK2 kinase activity alters neuronal autophagy by disrupting the axonal transport of autophagosomes
- PMID: 33765413
- PMCID: PMC8154747
- DOI: 10.1016/j.cub.2021.02.061
Increased LRRK2 kinase activity alters neuronal autophagy by disrupting the axonal transport of autophagosomes
Abstract
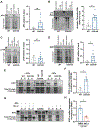
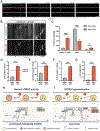
Parkinson's disease-causing mutations in the leucine-rich repeat kinase 2 (LRRK2) gene hyperactivate LRRK2 kinase activity and cause increased phosphorylation of Rab GTPases, important regulators of intracellular trafficking. We found that the most common LRRK2 mutation, LRRK2-G2019S, dramatically reduces the processivity of autophagosome transport in neurons in a kinase-dependent manner. This effect was consistent across an overexpression model, neurons from a G2019S knockin mouse, and human induced pluripotent stem cell (iPSC)-derived neurons gene edited to express the G2019S mutation, and the effect was reversed by genetic or pharmacological inhibition of LRRK2. Furthermore, LRRK2 hyperactivation induced by overexpression of Rab29, a known activator of LRRK2 kinase, disrupted autophagosome transport to a similar extent. Mechanistically, we found that hyperactive LRRK2 recruits the motor adaptor JNK-interacting protein 4 (JIP4) to the autophagosomal membrane, inducing abnormal activation of kinesin that we propose leads to an unproductive tug of war between anterograde and retrograde motors. Disruption of autophagosome transport correlated with a significant defect in autophagosome acidification, suggesting that the observed transport deficit impairs effective degradation of autophagosomal cargo in neurons. Our results robustly link increased LRRK2 kinase activity to defects in autophagosome transport and maturation, further implicating defective autophagy in the pathogenesis of Parkinson's disease.
Keywords: JIP4; LRRK2; Parkinson's disease; Rab29; autophagy; axonal transport.
Copyright © 2021 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare no competing interests.
Figures





Comment in
-
Neurobiology: A pathogenic tug of war.Curr Biol. 2021 May 24;31(10):R491-R493. doi: 10.1016/j.cub.2021.03.071. Curr Biol. 2021. PMID: 34033775 Free PMC article.
References
-
- Zimprich A, Biskup S, Leitner P, Lichtner P, Farrer M, Lincoln S, Kachergus J, Hulihan M, Uitti RJ, Calne DB, et al. (2004). Mutations in LRRK2 cause autosomaldominant parkinsonism with pleomorphic pathology. Neuron 44, 601–607. - PubMed
-
- Smith WW, Pei Z, Jiang H, Dawson VL, Dawson TM, and Ross CA (2006). Kinase activity of mutant LRRK2 mediates neuronal toxicity. Nat. Neurosci 9, 1231–1233. - PubMed
-
- Greggio E, Jain S, Kingsbury A, Bandopadhyay R, Lewis P, Kaganovich A, van der Brug MP, Beilina A, Blackinton J, Thomas KJ, et al. (2006). Kinase activity is required for the toxic effects of mutant LRRK2/dardarin. Neurobiol. Dis 23, 329–341. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

