A Randomized Trial of Tenapanor and Phosphate Binders as a Dual-Mechanism Treatment for Hyperphosphatemia in Patients on Maintenance Dialysis (AMPLIFY)
- PMID: 33766811
- PMCID: PMC8259655
- DOI: 10.1681/ASN.2020101398
A Randomized Trial of Tenapanor and Phosphate Binders as a Dual-Mechanism Treatment for Hyperphosphatemia in Patients on Maintenance Dialysis (AMPLIFY)
Abstract
Background: Hyperphosphatemia is associated with cardiovascular morbidity and mortality in patients receiving maintenance dialysis. It is unknown whether combining two therapies with different mechanisms of action-tenapanor, an inhibitor of paracellular phosphate absorption, and phosphate binders-is safe and effective for the management of hyperphosphatemia in patients receiving maintenance dialysis.
Methods: This double-blind phase 3 trial enrolled 236 patients undergoing maintenance dialysis with hyperphosphatemia (defined in this trial as serum phosphorus 5.5-10 mg/dl inclusive) despite receiving phosphate binder therapy (sevelamer, nonsevelamer, sevelamer plus nonsevelamer, or multiple nonsevelamer binders). These participants were randomly assigned to receive oral tenapanor 30 mg twice daily or placebo for 4 weeks. The primary efficacy end point was the change in serum phosphorus concentration from baseline to week 4.
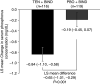
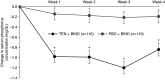
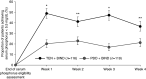
Results: Of the 236 randomized patients, 235 (99.6%) were included in the full analysis set; this included 116 in the tenapanor plus binder group and 119 in the placebo plus binder group. A total of 228 patients (96.6%) completed the 4-week treatment period. In the full analysis set (mean age 54.5 years, 40.9% women), patients treated with tenapanor plus binder achieved a larger mean change in serum phosphorus concentration from baseline to week 4 compared with placebo plus binder (-0.84 versus -0.19 mg/dl, P<0.001). Diarrhea was the most commonly reported adverse event, resulting in study drug discontinuation in four of 119 (3.4%) and two of 116 (1.7%) patients receiving tenapanor plus binder or placebo plus binder, respectively.
Conclusions: A dual-mechanism treatment using both tenapanor and phosphate binders improved control of hyperphosphatemia in patients undergoing maintenance dialysis compared with phosphate binders alone.
Clinical trial registry name and registration number: AMPLIFY, NCT03824587.
Keywords: FGF23; NHE3 dialysis; hyperphosphatemia; phosphate binders; phosphate uptake; phosphorus; tenapanor.
Copyright © 2021 by the American Society of Nephrology.
Figures

References
-
- Rastogi A, Bhatt N, Rossetti S, Beto J: Management of hyperphosphatemia in end-stage renal disease: A new paradigm. J Ren Nutr 31: 21–34, 2021. - PubMed
-
- Mizobuchi M, Towler D, Slatopolsky E: Vascular calcification: The killer of patients with chronic kidney disease. J Am Soc Nephrol 20: 1453–1464, 2009. - PubMed
-
- Sigrist MK, Taal MW, Bungay P, McIntyre CW: Progressive vascular calcification over 2 years is associated with arterial stiffening and increased mortality in patients with stages 4 and 5 chronic kidney disease. Clin J Am Soc Nephrol 2: 1241–1248, 2007. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

