Efficacy and Safety of Single Oral Dosing of Secnidazole for Trichomoniasis in Women: Results of a Phase 3, Randomized, Double-Blind, Placebo-Controlled, Delayed-Treatment Study
- PMID: 33768237
- PMCID: PMC8442793
- DOI: 10.1093/cid/ciab242
Efficacy and Safety of Single Oral Dosing of Secnidazole for Trichomoniasis in Women: Results of a Phase 3, Randomized, Double-Blind, Placebo-Controlled, Delayed-Treatment Study
Abstract
Background: Trichomonas vaginalis is the most prevalent nonviral sexually transmitted infection. We evaluated the efficacy and safety of secnidazole vs placebo in women with trichomoniasis.
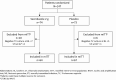
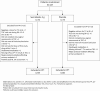
Methods: Women with trichomoniasis, confirmed by a positive T. vaginalis culture, were randomized to single-dose oral secnidazole 2 g or placebo. The primary endpoint was microbiological test of cure (TOC) by culture 6-12 days after dosing. At the TOC visit, participants were given the opposite treatment. They were followed for resolution of infection afterward and offered treatment at subsequent visits, if needed. Fifty patients per group (N = 100) provided approximately 95% power to detect a statistically significant difference between treatment groups.
Results: Between April 2019 and March 2020, 147 women enrolled at 10 sites in the United States. The modified intention-to-treat (mITT) population included 131 randomized patients (secnidazole, n = 64; placebo, n = 67). Cure rates were significantly higher in the secnidazole vs placebo group for the mITT population (92.2% [95% confidence interval {CI}: 82.7%-97.4%] vs 1.5% [95% CI: .0%-8.0%]) and for the per-protocol population (94.9% [95% CI: 85.9%-98.9%] vs 1.7% [95% CI: .0%-8.9%]). Cure rates were 100% (4/4) in women with human immunodeficiency virus (HIV) and 95.2% (20/21) in women with bacterial vaginosis (BV). Secnidazole was generally well tolerated. The most frequently reported treatment-emergent adverse events (TEAEs) were vulvovaginal candidiasis and nausea (each 2.7%). No serious TEAEs were observed.
Conclusions: A single oral 2 g dose of secnidazole was associated with significantly higher microbiological cure rates vs placebo, supporting a role for secnidazole in treating women with trichomoniasis, including those with HIV and/or BV.
Clinical trials registration: NCT03935217.
Keywords: Trichomonas vaginalis; secnidazole; trichomoniasis; women.
© The Author(s) 2021. Published by Oxford University Press for the Infectious Diseases Society of America.
Figures
Similar articles
-
Efficacy of single-dose oral secnidazole for the treatment of trichomoniasis in women co-infected with trichomoniasis and bacterial vaginosis: a post hoc subgroup analysis of phase 3 clinical trial data.BMJ Open. 2023 Aug 7;13(8):e072071. doi: 10.1136/bmjopen-2023-072071. BMJ Open. 2023. PMID: 37550019 Free PMC article. Clinical Trial.
-
A phase-3, double-blind, placebo-controlled study of the effectiveness and safety of single oral doses of secnidazole 2 g for the treatment of women with bacterial vaginosis.Am J Obstet Gynecol. 2017 Dec;217(6):678.e1-678.e9. doi: 10.1016/j.ajog.2017.08.017. Epub 2017 Sep 1. Am J Obstet Gynecol. 2017. PMID: 28867602 Clinical Trial.
-
Secnidazole for Trichomoniasis in Women and Men.Sex Med Rev. 2022 Apr;10(2):255-262. doi: 10.1016/j.sxmr.2021.12.004. Epub 2022 Feb 10. Sex Med Rev. 2022. PMID: 35153156 Free PMC article. Review.
-
Secnidazole Treatment of Bacterial Vaginosis: A Randomized Controlled Trial.Obstet Gynecol. 2017 Aug;130(2):379-386. doi: 10.1097/AOG.0000000000002135. Obstet Gynecol. 2017. PMID: 28697102 Clinical Trial.
-
Secnidazole for treatment of bacterial vaginosis: a systematic review.BMC Womens Health. 2019 Oct 21;19(1):121. doi: 10.1186/s12905-019-0822-2. BMC Womens Health. 2019. PMID: 31638955 Free PMC article.
Cited by
-
Secnidazole: a treatment for trichomoniasis in adolescents and adults.Expert Rev Anti Infect Ther. 2022 Aug;20(8):1067-1076. doi: 10.1080/14787210.2022.2080656. Epub 2022 Jun 8. Expert Rev Anti Infect Ther. 2022. PMID: 35642509 Free PMC article.
-
Identification of Trichomonas vaginalis 5-Nitroimidazole Resistance Targets.Pathogens. 2023 May 10;12(5):692. doi: 10.3390/pathogens12050692. Pathogens. 2023. PMID: 37242362 Free PMC article.
-
Detection of Trichomonas vaginalis by Allplex™ STI Essential Assay (Seegene) in clinical samples from the Brazilian public health system users.Eur J Clin Microbiol Infect Dis. 2024 Jan;43(1):167-170. doi: 10.1007/s10096-023-04694-7. Epub 2023 Nov 2. Eur J Clin Microbiol Infect Dis. 2024. PMID: 37917224
-
Trichomonas vaginalis Virus: Current Insights and Emerging Perspectives.Viruses. 2025 Jun 26;17(7):898. doi: 10.3390/v17070898. Viruses. 2025. PMID: 40733518 Free PMC article. Review.
-
Effect of Trichomonacide 6-Nitro-1H-benzimidazole Derivative Compounds on Expression Level of Metabolic Genes in Trichomonas vaginalis.Int J Mol Sci. 2024 Apr 22;25(8):4568. doi: 10.3390/ijms25084568. Int J Mol Sci. 2024. PMID: 38674152 Free PMC article.
References
-
- McClelland RS, Sangare L, Hassan WM, et al. . Infection with trichomonas vaginalis increases the risk of HIV-1 acquisition. J Infect Dis 2007; 195:698–702. - PubMed
-
- Committee on Practice Bulletins-Gynecology. Vaginitis in nonpregnant patients: ACOG practice bulletin, number 215. Obstet Gynecol 2020; 135:e1–17. - PubMed
-
- Mielczarek E, Blaszkowska J. Trichomonas vaginalis: pathogenicity and potential role in human reproductive failure. Infection 2016; 44:447–58. - PubMed
-
- Silver BJ, Guy RJ, Kaldor JM, Jamil MS, Rumbold AR. Trichomonas vaginalis as a cause of perinatal morbidity: a systematic review and meta-analysis. Sex Transm Dis 2014; 41:369–76. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

