Drug-Free Enzyme-Based Bactericidal Nanomotors against Pathogenic Bacteria
- PMID: 33769023
- PMCID: PMC8478280
- DOI: 10.1021/acsami.1c00986
Drug-Free Enzyme-Based Bactericidal Nanomotors against Pathogenic Bacteria
Abstract
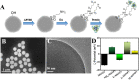
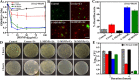
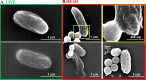
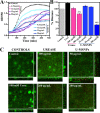
The low efficacy of current conventional treatments for bacterial infections increases mortality rates worldwide. To alleviate this global health problem, we propose drug-free enzyme-based nanomotors for the treatment of bacterial urinary-tract infections. We develop nanomotors consisting of mesoporous silica nanoparticles (MSNPs) that were functionalized with either urease (U-MSNPs), lysozyme (L-MSNPs), or urease and lysozyme (M-MSNPs), and use them against nonpathogenic planktonic Escherichia coli. U-MSNPs exhibited the highest bactericidal activity due to biocatalysis of urea into NaHCO3 and NH3, which also propels U-MSNPs. In addition, U-MSNPs in concentrations above 200 μg/mL were capable of successfully reducing 60% of the biofilm biomass of a uropathogenic E. coli strain. This study thus provides a proof-of-concept, demonstrating that enzyme-based nanomotors are capable of fighting infectious diseases. This approach could potentially be extended to other kinds of diseases by selecting appropriate biomolecules.
Keywords: E. coli; biofilms; enzymatic nanomotors; infections; nanomachines; self-propulsion.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

