Fluorinated Azobenzenes Switchable with Red Light
- PMID: 33769596
- PMCID: PMC8252058
- DOI: 10.1002/chem.202005486
Fluorinated Azobenzenes Switchable with Red Light
Abstract
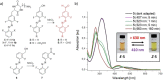
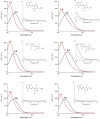
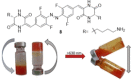
Molecular photoswitches triggered with red or NIR light are optimal for photomodulation of complex biological systems, including efficient penetration of the human body for therapeutic purposes ("therapeutic window"). Yet, they are rarely reported, and even more rarely functional under aqueous conditions. In this work, fluorinated azobenzenes are shown to exhibit efficient E→Z photoisomerization with red light (PSS660nm >75 % Z) upon conjugation with unsaturated substituents. Initially demonstrated for aldehyde groups, this effect was also observed in a more complex structure by incorporating the chromophore into a cyclic dipeptide with propensity for self-assembly. Under physiological conditions, the latter molecule formed a supramolecular material that reversibly changed its viscosity upon irradiation with red light. Our observation can lead to design of new photopharmacology agents or phototriggered materials for in vivo use.
Keywords: azobenzene; photoswitches; red-light photoisomerization; therapeutic window.
© 2021 The Authors. Published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

