Optimization of ether and aniline based inhibitors of lactate dehydrogenase
- PMID: 33771585
- PMCID: PMC8113097
- DOI: 10.1016/j.bmcl.2021.127974
Optimization of ether and aniline based inhibitors of lactate dehydrogenase
Abstract
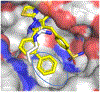
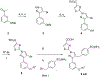
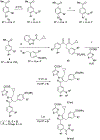
Lactate dehydrogenase (LDH) is a critical enzyme in the glycolytic metabolism pathway that is used by many tumor cells. Inhibitors of LDH may be expected to inhibit the metabolic processes in cancer cells and thus selectively delay or inhibit growth in transformed versus normal cells. We have previously disclosed a pyrazole-based series of potent LDH inhibitors with long residence times on the enzyme. Here, we report the elaboration of a new subseries of LDH inhibitors based on those leads. These new compounds potently inhibit both LDHA and LDHB enzymes, and inhibit lactate production in cancer cell lines.
Keywords: Cancer metabolism; LDH inhibitor; Medicinal chemistry; Structure activity relationships.
Copyright © 2021 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Declaration of interests
The authors declare the following financial interests/personal relationships which may be considered as potential competing interests:
Compounds reported in this manuscript are also exemplified in patent applications that have been licensed. Under certain circumstances, co-authors on this manuscript may receive royalties or other payments through their respective employers.
Figures

References
-
- Warburg O; Posener K; Negelein E Biochem. Z 1924, 152, 319–344.
-
- Rong Y; Wu W; Ni X; Kuang T; Jin D; Wang D; Lou W Tumour Biol. 2013, 34, 1523–1530. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Chemical Information
Miscellaneous

