Macrophage inhibitory cytokine-1 induced by a high-fat diet promotes prostate cancer progression by stimulating tumor-promoting cytokine production from tumor stromal cells
- PMID: 33773090
- PMCID: PMC8118591
- DOI: 10.1002/cac2.12137
Macrophage inhibitory cytokine-1 induced by a high-fat diet promotes prostate cancer progression by stimulating tumor-promoting cytokine production from tumor stromal cells
Abstract
Background: Recent studies have indicated that a high-fat diet (HFD) and/or HFD-induced obesity may influence prostate cancer (PCa) progression, but the role of HFD in PCa microenvironment is unclear. This study aimed to delineate the molecular mechanisms of PCa progression under HFD milieus and define the stromal microenvironment focusing on macrophage inhibitory cytokine-1 (MIC-1) activation.
Methods: We investigated the effects of HFD on PCa stromal microenvironment and MIC-1 signaling activation using PC-3M-luc-C6 PCa model mice fed with HFD or control diet. Further, we explored the effect of periprostatic adipocytes derived from primary PCa patients on activation and cytokine secretion of prostate stromal fibroblasts. Expression patterns and roles of MIC-1 signaling on human PCa stroma activation and progression were also investigated.
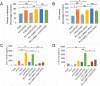
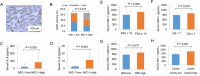
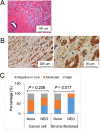
Results: HFD stimulated PCa cell growth and invasion as a result of upregulated MIC-1 signaling and subsequently increased the secretion of interleukin (IL)-8 and IL-6 from prostate stromal fibroblasts in PC-3M-luc-C6 PCa mouse model. In addition, periprostatic adipocytes directly stimulated MIC-1 production from PC-3 cells and IL-8 secretion in prostate stromal fibroblasts through the upregulation of adipose lipolysis and free fatty acid release. The increased serum MIC-1 was significantly correlated with human PCa stroma activation, high serum IL-8, IL-6, and lipase activity, advanced PCa progression, and high body mass index of the patients. Glial-derived neurotrophic factor receptor α-like (GFRAL), a specific receptor of MIC-1, was highly expressed in both cytoplasm and membrane of PCa cells and surrounding stromal fibroblasts, and the expression level was decreased by androgen deprivation therapy and chemotherapy.
Conclusion: HFD-mediated activation of the PCa stromal microenvironment through metabolically upregulated MIC-1 signaling by increased available free fatty acids may be a critical mechanism of HFD and/or obesity-induced PCa progression.
Keywords: high-fat diet; macrophage inhibitory cytokine-1; metabolism; prostate cancer; tumor microenvironment.
© 2021 The Authors. Cancer Communications published by John Wiley & Sons Australia, Ltd. on behalf of Sun Yat-sen University Cancer Center.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures




References
-
- Calle EE, Rodriguez C, Walker‐Thurmond K, Thun MJ. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of U.S. adults. N Engl J Med. 2003;348(17):1625‐38. - PubMed
-
- Narita S, Tsuchiya N, Saito M, Inoue T, Kumazawa T, Yuasa T, et al. Candidate genes involved in enhanced growth of human prostate cancer under high fat feeding identified by microarray analysis. The Prostate. 2008;68(3):321‐35. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

