The Bioequivalence of Emulsified Isoflurane With a New Formulation of Emulsion: A Single-Center, Single-Dose, Double-Blinded, Randomized, Two-Period Crossover Study
- PMID: 33776768
- PMCID: PMC7988084
- DOI: 10.3389/fphar.2021.626307
The Bioequivalence of Emulsified Isoflurane With a New Formulation of Emulsion: A Single-Center, Single-Dose, Double-Blinded, Randomized, Two-Period Crossover Study
Abstract
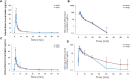
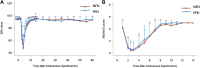
Background: Emulsified isoflurane is a novel intravenous general anesthetic obtained by encapsulating isoflurane molecules into emulsion. The formulation of emulsion has been improved according to the latest regulations of the China Food and Drug Administration. This study was designed to compare the bioequivalence of the new and previous formulation emulsion of isoflurane. Methods: In a single-center, single-dose, double-blinded, randomized, two-period crossover study, healthy volunteers received intravenous injection of 30 mg/kg of isoflurane with either previous formulation of emulsion isoflurane (PFEI) or new formulation of emulsion isoflurane (NFEI). Arterial and venous blood samples were obtained for geometric mean test/reference ratios of Cmax, AUC0-t, and AUC0-∞, as well as their 90% confidence interval (CI90) as the primary outcome. The secondary outcomes were safety measurements such as vital signs, 12-lead electrocardiography, adverse effects, and laboratory tests; and anesthesia efficacy was assessed by Modified Observer's Assessment of Alertness/Sedation (MOAA/S) score, bispectral index (BIS), and loss/recovery of eyelash reflex. Results: 24 subjects were eligible, of which 21 completed the whole experiment (NFEI n = 21, PFEI n = 23). Arterial geometric mean test/reference ratios of Cmax, AUC0-t, and AUC0-∞ were 104.50% (CI90 92.81%-117.65%), 108.23% (94.51%-123.96%), and 106.53% (93.94%∼120.80%), respectively. The most commonly seen adverse effects for NFEI and PFEI were injection pain (38.1% vs. 34.8%), hypotension (19.0% vs. 13.0%), apnea (14.3% vs. 17.4%), and upper airway obstruction (14.3% vs. 13.0%). No severe adverse effect was observed. The effectiveness of general anesthesia was similar between the two formulations. Conclusion: The CI90 of Cmax, AUC0-t, AUC0-∞, NFEI, and PFEI were within the range of 80%-125%, suggesting bioequivalence between NFEI and PFEI. The safety and anesthesia effectiveness were also similar.
Keywords: AUC0-t; AUC0-∞; Cmax; bioequivalence; emulsified isoflurane.
Copyright © 2021 Yang, Yin, Huang, Zhang, Zhang, Sun, Liu, Wang, Yang, Tan, Ye and Liu.
Conflict of interest statement
MZ and XZ are employees of Yichang Humanwell Pharmaceutical Co., Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Bienvenu B., Aouba A., Gottenberg J. E., Verstuyft C. (2017). Single-center, single-dose, open-label, randomized, two-period crossover study on the bioavailability of methotrexate administered using a novel prefilled, needle-free delivery system. Curr. Med. Res. Opin. 33 (4), 605–611. 10.1080/03007995.2016.1263611 - DOI - PubMed
-
- Davanço M. G., Meulman J., Guzmán M. R. P., Palomino D. M. H., Tuiran C. M. G., Duarte F. G., et al. (2019). A randomized, single-dose, two-sequence, two-period, crossover study to assess the bioequivalence between two formulations of clonazepam tablet in healthy subjects. Drug Dev. Ind. Pharm. 45 (12), 1982–1987. 10.1080/03639045.2019.1689994 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

