Zi Shen Decoction Inhibits Growth and Metastasis of Lung Cancer via Regulating the AKT/GSK-3 β/ β-Catenin Pathway
- PMID: 33777320
- PMCID: PMC7969097
- DOI: 10.1155/2021/6685282
Zi Shen Decoction Inhibits Growth and Metastasis of Lung Cancer via Regulating the AKT/GSK-3 β/ β-Catenin Pathway
Abstract
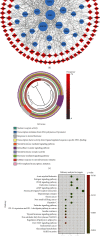
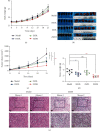
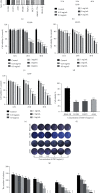
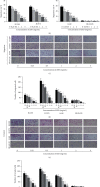
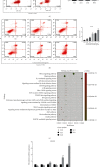
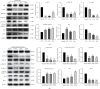
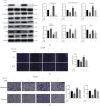
Lung cancer has become the leading cause of cancer-related death worldwide. Oxidative stress plays important roles in the pathogenesis of lung cancer. Many natural products show antioxidative activities in cancer treatment. Zi Shen decoction (ZSD) is a classic prescription for the treatment of lung disease. However, its effect on lung cancer lacks evidence-based efficacy. In this study, we investigated the anticancer effects of ZSD on lung cancer in vivo and in vitro. Our results showed that oral administration of ZSD suppressed the Lewis lung cancer (LLC) growth in a subcutaneous allograft model and promoted necrosis and inflammatory cell infiltration in the tumor tissues. Furthermore, ZSD not only inhibited tumor cell proliferation and migration but also induced cell apoptosis in lung cancer cells. PI3K/AKT signaling is well characterized in response to oxidative stress. The bioinformatics analysis and western blot assays suggested that ZSD decreased the enzyme activity of PI3K and AKT in vivo and in vitro. We also found that the AKT/GSK-3β/β-catenin pathway medicated anticancer effect of ZSD in lung cancer cells. In conclusion, we demonstrate for the first time that ZSD possesses antitumor properties, highlighting its potential use as an alternative strategy or adjuvant treatment for lung cancer therapy.
Copyright © 2021 Yanxia Ma et al.
Conflict of interest statement
All the authors have read and approved the final manuscript, and there is no known competing interest.
Figures
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

