Genetic Fusion of Transacting Activator of Transcription Peptide to Cyclized Green Fluorescence Protein Improves Stability, Intracellular Delivery, and Tumor Retention
- PMID: 33778304
- PMCID: PMC7992142
- DOI: 10.1021/acsomega.1c00532
Genetic Fusion of Transacting Activator of Transcription Peptide to Cyclized Green Fluorescence Protein Improves Stability, Intracellular Delivery, and Tumor Retention
Abstract
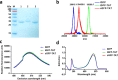
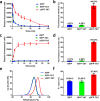
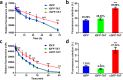
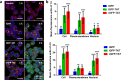
Therapeutic proteins such as enzymes, hormones, and cytokines suffer from poor stability, inefficient cellular penetration, and rapid clearance from circulation. Conjugation with polymers (such as poly(ethylene glycol)) and fusion with long-acting proteins (such as albumin and Fc fragments) have been utilized to partially address the delivery issues, but these strategies require the introduction of new macromolecular substances, resulting in potential immunogenicity and toxicity. Herein, we report an easy strategy to increase the intracellular delivery efficiency and stability of proteins by combining of sortase-mediated protein cyclization and cell-penetrating peptide (CPP)-mediated intracellular delivery. We, for the first time, genetically constructed a green fluorescence protein (GFP) fused with a CPP, a transacting activator of transcription (TAT) peptide, at its C-terminus for intracellular internalization, and two sortase recognition sequences, pentaglycine and LPETG, at its N- and C-termini for cyclization. Notably, the cyclized GFP-TAT (cGFP-TAT) not only highly retained the photophysical properties of the protein but also significantly improved the in vitro stability compared with the native linear GFP (lGFP) and linear TAT peptide-fused GFP (lGFP-TAT).Moreover, cGFP-TAT showed better cellular internalization ability compared with lGFP. In C26 tumor-inoculated mice, cGFP-TAT exhibited enhanced in vivo tumor retention, with increases of 7.79- and 6.52-fold relative to lGFP and lGFP-TAT in tumor retention 3 h after intratumor administration. This proof-of-concept study has provided an easy strategy to increase the in vitro stability, intracellular delivery efficiency, and in vivo tumor retention of GFP, which would be applicable to numerous therapeutic proteins and peptides for clinical practice.
© 2021 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures


Similar articles
-
Site-specific in situ growth of a cyclized protein-polymer conjugate with improved stability and tumor retention.Biomaterials. 2015 Apr;47:13-9. doi: 10.1016/j.biomaterials.2015.01.002. Epub 2015 Feb 2. Biomaterials. 2015. PMID: 25682156
-
Can the Cellular Internalization of Cargo Proteins Be Enhanced by Fusing a Tat Peptide in the Center of Proteins? A Fluorescence Study.J Pharm Sci. 2018 Mar;107(3):879-886. doi: 10.1016/j.xphs.2017.11.002. Epub 2017 Nov 11. J Pharm Sci. 2018. PMID: 29133235
-
Enhancement of bioactivity, thermal stability and tumor retention by self-fused concatenation of green fluorescent protein.Biochem Biophys Rep. 2021 Aug 24;28:101112. doi: 10.1016/j.bbrep.2021.101112. eCollection 2021 Dec. Biochem Biophys Rep. 2021. PMID: 34485712 Free PMC article.
-
CPP-mediated protein delivery in a noncovalent form: proof-of-concept for percutaneous and intranasal delivery.Protein Pept Lett. 2014;21(11):1129-36. doi: 10.2174/0929866521666140807121903. Protein Pept Lett. 2014. PMID: 25106905 Review.
-
In vitro and in vivo delivery of therapeutic proteins using cell penetrating peptides.Peptides. 2017 Jan;87:50-63. doi: 10.1016/j.peptides.2016.11.011. Epub 2016 Nov 22. Peptides. 2017. PMID: 27887988 Review.
Cited by
-
Applications of cell penetrating peptide-based drug delivery system in immunotherapy.Front Immunol. 2025 Jan 22;16:1540192. doi: 10.3389/fimmu.2025.1540192. eCollection 2025. Front Immunol. 2025. PMID: 39911386 Free PMC article. Review.
-
Sortase-mediated labeling: Expanding frontiers in site-specific protein functionalization opens new research avenues.Curr Opin Chem Biol. 2024 Jun;80:102443. doi: 10.1016/j.cbpa.2024.102443. Epub 2024 Mar 19. Curr Opin Chem Biol. 2024. PMID: 38503199 Free PMC article. Review.
References
-
- Liu X.; Hu J.; Wang Z.; Xu Z.; Gao W. Polypeptides and Engineered Proteins. Acs. Sym. Ser. 2017, 1252, 93–127. 10.1021/bk-2017-1252.ch006. - DOI
LinkOut - more resources
Full Text Sources
Other Literature Sources

