Elective Colectomy in a Patient with Active Ulcerative Colitis and Metastatic Melanoma Enabling Successful Treatment with Immune Checkpoint Inhibitors
- PMID: 33778814
- PMCID: PMC7993656
Elective Colectomy in a Patient with Active Ulcerative Colitis and Metastatic Melanoma Enabling Successful Treatment with Immune Checkpoint Inhibitors
Abstract
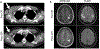
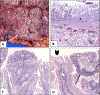
Checkpoint inhibitor immunotherapy has significantly advanced treatment of a growing number of advanced malignancies. A consequences of immune system activation that leads to tumor cell destruction by checkpoint inhibitor therapy is the development of immune-related adverse events, some of which can be life threatening. There are limited data on the use of checkpoint inhibitor therapy in patients with preexisting autoimmunity owing to concerns that underlying autoimmune disease may be exacerbated by checkpoint inhibitor treatment. Decisions to treat these patients are made after careful consideration of the risks and benefits of treatment. We describe a patient with active and severe ulcerative colitis with metastatic melanoma who underwent elective colectomy prior to initiation of anti-PD-1 and anti-CTLA-4. The patient had excellent tumor response without flare of his ulcerative colitis suggesting that in select patients with high-risk inflammatory bowel disease, elective colectomy may be an effective treatment option.
Keywords: Checkpoint; Colitis; Complications; Immunotherapy; Inflammatory bowel disease.
Conflict of interest statement
Conflicts of interest H. Kluger reports research grants from Merck, Bristol-Myers Squibb, and Apexigen during the conduct of the study, and personal fees from Corvus, Nektar, Biodesix, Roche-Genetech, Pfizer, Iovance, Immunocore, and Celldex, Array Biopharma, Bristol-Myers Squibb, Clinigen and Merck, outside of the submitted work.
Figures

Similar articles
-
Severe autoimmune hemolytic anemia following immunotherapy with checkpoint inhibitors in two patients with metastatic melanoma: a case report.Front Immunol. 2024 Mar 20;15:1342845. doi: 10.3389/fimmu.2024.1342845. eCollection 2024. Front Immunol. 2024. PMID: 38571955 Free PMC article.
-
Immune checkpoint inhibitor refractory colitis leading to total colectomy in a melanoma patient.Acta Gastroenterol Belg. 2023 Apr-Jun;86(2):371-373. doi: 10.51821/86.2.10400. Acta Gastroenterol Belg. 2023. PMID: 37428174
-
Rapid complete response of metastatic melanoma in a patient undergoing ipilimumab immunotherapy in the setting of active ulcerative colitis.J Immunother Cancer. 2015 May 19;3:19. doi: 10.1186/s40425-015-0064-2. eCollection 2015. J Immunother Cancer. 2015. PMID: 25992290 Free PMC article.
-
The Risk of Diarrhea and Colitis in Patients With Advanced Melanoma Undergoing Immune Checkpoint Inhibitor Therapy: A Systematic Review and Meta-Analysis.J Immunother. 2018 Apr;41(3):101-108. doi: 10.1097/CJI.0000000000000213. J Immunother. 2018. PMID: 29401166
-
The Next Immune-Checkpoint Inhibitors: PD-1/PD-L1 Blockade in Melanoma.Clin Ther. 2015 Apr 1;37(4):764-82. doi: 10.1016/j.clinthera.2015.02.018. Epub 2015 Mar 29. Clin Ther. 2015. PMID: 25823918 Free PMC article. Review.
Cited by
-
Checkpoint Inhibitor-Induced Colitis: An Update.Biomedicines. 2023 May 22;11(5):1496. doi: 10.3390/biomedicines11051496. Biomedicines. 2023. PMID: 37239166 Free PMC article. Review.
References
-
- Postow MA, Sidlow R, Hellmann MD (2018) Immune-related adverse events associated with immune checkpoint blockade. N Engl J Med 378: 158–168. - PubMed
-
- Soularue E, Lepage P, Colombel JF, Coutzac C, Faleck D, et al. (2018) Enterocolitis due to immune checkpoint inhibitors: A systematic review. Gut 67: 2056–2067. - PubMed
-
- Brahmer JR, Lacchetti C, Thompson JA (2018) Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: American society of clinical oncology clinical practice guideline summary. J Oncol Pract 14: 247–249. - PubMed
-
- Abdel-Wahab N, Shah M, Lopez-Olivo MA, Suarez-Almazor ME (2018) Use of immune checkpoint inhibitors in the treatment of patients with cancer and preexisting autoimmune disease: A systematic review. Ann Intern Med 168: 121–130. - PubMed
-
- Menzies AM, Johnson DB, Ramanujam S, Atkinson VG, Wong ANM, et al. (2017) Anti-PD-1 therapy in patients with advanced melanoma and preexisting autoimmune disorders or major toxicity with ipilimumab. Ann Oncol 28: 368–376. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
