Fuzzy logic and Lyapunov-based non-linear controllers for HCV infection
- PMID: 33780147
- PMCID: PMC8675797
- DOI: 10.1049/syb2.12014
Fuzzy logic and Lyapunov-based non-linear controllers for HCV infection
Abstract
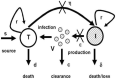
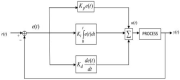
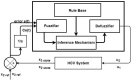
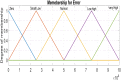
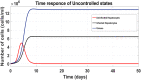
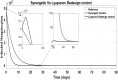
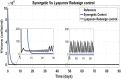
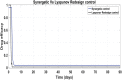
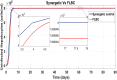
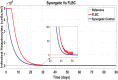
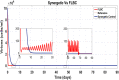
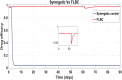
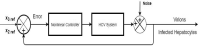
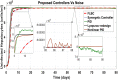
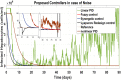
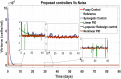
Hepatitis C is the liver disease caused by the Hepatitis C virus (HCV) which can lead to serious health problems such as liver cancer. In this research work, the non-linear model of HCV having three state variables (uninfected hepatocytes, infected hepatocytes and virions) and two control inputs has been taken into account, and four non-linear controllers namely non-linear PID controller, Lyapunov Redesign controller, Synergetic controller and Fuzzy Logic-Based controller have been proposed to control HCV infection inside the human body. The controllers have been designed for the anti-viral therapy in order to control the amount of uninfected hepatocytes to the desired safe limit and to track the amount of infected hepatocytes and virions to their reference value which is zero. One control input is the Pegylated interferon (peg-IFN-α) which acts in reducing the infected hepatocytes and the other input is ribavirin which blocks the production of virions. By doing so, the uninfected hepatocytes increase and achieve the required safe limit. Lyapunov stability analysis has been used to prove the stability of the whole system. The comparative analysis of the proposed nonlinear controllers using MATLAB/Simulink have been done with each other and with linear PID. These results depict that the infected hepatocytes and virions are reduced to the desired level, enhancing the rate of sustained virologic response (SVR) and reducing the treatment period as compared with previous strategies introduced in the literature.
© 2021 The Authors. IET Systems Biology published by John Wiley & Sons Ltd on behalf of The Institution of Engineering and Technology.
Figures


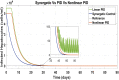
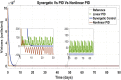
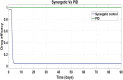
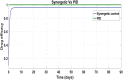



Similar articles
-
Early discontinuation of ribavirin in HCV-2 and HCV-3 patients responding to Peg-interferon alpha-2a and ribavirin.J Viral Hepat. 2009 Jan;16(1):28-35. doi: 10.1111/j.1365-2893.2008.01044.x. Epub 2008 Aug 28. J Viral Hepat. 2009. PMID: 18761603 Clinical Trial.
-
Virological response in patients with hepatitis C virus genotype 1b and a high viral load: impact of peginterferon-alpha-2a plus ribavirin dose reductions and host-related factors.Clin Drug Investig. 2008;28(1):9-16. doi: 10.2165/00044011-200828010-00002. Clin Drug Investig. 2008. PMID: 18081356
-
Boceprevir plus pegylated interferon/ribavirin to re-treat hepatitis C virus genotype 1 in HIV-HCV co-infected patients: final results of the Spanish BOC HIV-HCV Study.Int J Infect Dis. 2016 Dec;53:46-51. doi: 10.1016/j.ijid.2016.10.028. Epub 2016 Nov 1. Int J Infect Dis. 2016. PMID: 27815225 Clinical Trial.
-
Consensus guidelines for the management of hepatitis C infection.Saudi Med J. 2003 Jul;24 Suppl 2:S99-118. Saudi Med J. 2003. PMID: 25121186
-
Options for treatment of hepatitis C in HIV-infected persons.J Hepatol. 2006;44(1 Suppl):S40-3. doi: 10.1016/j.jhep.2005.11.011. Epub 2005 Nov 28. J Hepatol. 2006. PMID: 16356578 Review.
References
-
- Choo, Q.‐L. , et al.: Isolation of a cdna clone derived from a blood‐borne non‐a, non‐b viral hepatitis genome. Science. 244(4902), 359–362 (1989) - PubMed
-
- Organization, W.H. , et al.: Global hepatitis report 2017. World Health Organization; (2017). https://scholar.googleusercontent.com/scholar?q=cache:w1LIjp_UH7kJ:schol...
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

