Baseline Testosterone Levels in Men with Clinically Localized High-Risk Prostate Cancer Treated with Radical Prostatectomy with or without Neoadjuvant Chemohormonal Therapy (Alliance)
- PMID: 33780276
- PMCID: PMC8273099
- DOI: 10.1097/JU.0000000000001716
Baseline Testosterone Levels in Men with Clinically Localized High-Risk Prostate Cancer Treated with Radical Prostatectomy with or without Neoadjuvant Chemohormonal Therapy (Alliance)
Abstract
Purpose: Men with low serum testosterone at the time of prostate cancer diagnosis are frequently considered to have more aggressive disease. We examined treatment outcomes in men with clinically localized high-risk cancer to determine if baseline testosterone level identified men at higher risk for cancer progression after treatment.
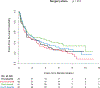
Materials and methods: Alliance/CALGB 90203 randomized men with clinically localized high-risk prostate cancer to radical prostatectomy alone or neoadjuvant chemohormonal therapy and radical prostatectomy. Men with available baseline testosterone levels who had not received androgen deprivation prior to study enrollment were studied (656). Testosterone level was examined as a continuous variable, as quartiles, and separately in men with an absolute testosterone level above/below 150 ng/dl. Outcomes evaluated were overall survival and event-free survival with events defined by biochemical recurrence, secondary treatment, prostate cancer metastasis, and death.
Results: We were unable to demonstrate a difference between baseline serum testosterone level measured as a continuous variable, as quartiles, or as a dichotomous variable (above/below 150 ng/dl) with the outcomes measured. This finding was observed in both arms of the study.
Conclusions: Baseline serum testosterone level did not predict outcomes in men with clinically localized high-risk prostate cancer treated with radical prostatectomy alone or neoadjuvant chemohormonal therapy and radical prostatectomy.
Keywords: neoadjuvant therapy; prostatectomy; prostatic neoplasms; testosterone.
Figures



References
-
- Yamamoto S, Yonese J, Kawakami S, et al. : Preoperative serum testosterone level as an independent predictor of treatment failure following radical prostatectomy. Eur Urol 2007; 52: 696. - PubMed
-
- Xylinas E, Ploussard G, Durand X, et al. : Low pretreatment total testosterone (< 3 ng/mL) predicts extraprostatic disease in prostatectomy specimens from patients with preoperative localized prostate cancer. BJU Int 2011; 107: 1400. - PubMed
-
- Massengill JC, Sun L, Moul JW, et al. : Pretreatment total testosterone level predicts pathological stage in patients with localized prostate cancer treated with radical prostatectomy. J Urol 2003; 169: 1670. - PubMed
-
- Porcaro AB, Tafuri A, Sebben M, et al. : Positive association between preoperative total testosterone levels and risk of positive surgical margins by prostate cancer: results in 476 consecutive patients treated only by radical prostatectomy. Urol Int 2018; 101: 38. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

