MASP-1 and MASP-3 Bind Directly to Aspergillus fumigatus and Promote Complement Activation and Phagocytosis
- PMID: 33780946
- PMCID: PMC8460983
- DOI: 10.1159/000514546
MASP-1 and MASP-3 Bind Directly to Aspergillus fumigatus and Promote Complement Activation and Phagocytosis
Abstract
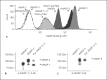
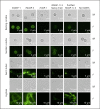
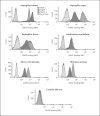
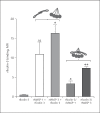
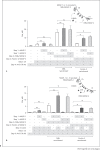
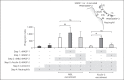
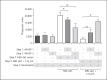
Activation of the complement system is mediated by the interaction between pathogens and pattern recognition molecules (PRMs); mannose-binding lectin (MBL), ficolins, and collectin-10/-11 from the lectin pathway and C1q from the classical pathway. Lectin pathway activation specifically depends on proteases named MBL-associated serine proteases (MASPs) that are found in complexes with PRMs. In this study, we hypothesize that MASPs can recognize selected pathogens independently of PRMs. Using different clinical strains of opportunistic fungi, we have observed that MASPs directly recognize certain fungal pathogens in a way that can facilitate complement activation. Among these were Aspergillus fumigatus - a dangerous pathogen, especially for immunocompromised patients. In flow cytometry and fluorescence microscopy, we found that MASP-1 and -3 bound to all A. fumigatus growth stages (conidia, germ tubes, and hyphae), whereas rMASP-2 and the nonproteolytic rMAP-1 did not. Bound rMASPs could recruit rMBL and rficolin-3 to A. fumigatus conidia in a nonclassical manner and activate complement via rMASP-2. In experiments using recombinant and purified components, rMASP-1 increased the neutrophilic phagocytosis of conidia. In serum where known complement activation pathways were blocked, phagocytosis could be mediated by rMASP-3. We have encountered an unknown pathway for complement activation and found that MASP-1 and MASP-3 have dual functions as enzymes and as PRMs.
Keywords: Aspergillosis; Fungi; Lectin pathway; Mannose-binding lectin-associated serine protease; Pattern recognition molecules.
© 2021 The Author(s). Published by S. Karger AG, Basel.
Conflict of interest statement
The authors have no conflict of interest to declare.
Figures



References
-
- Garred P, Genster N, Pilely K, Bayarri-Olmos R, Rosbjerg A, Ma YJ, et al. A journey through the lectin pathway of complement-MBL and beyond. Immunol Rev. 2016;274((1)):74–97. - PubMed
-
- Matsushita M, Thiel S, Jensenius JC, Terai I, Fujita T. Proteolytic activities of two types of mannose-binding lectin-associated serine protease. J Immunol. 2000;165((5)):2637–42. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

