Transplantation of human induced pluripotent stem cell-derived neural crest cells for corneal endothelial regeneration
- PMID: 33781330
- PMCID: PMC8008577
- DOI: 10.1186/s13287-021-02267-z
Transplantation of human induced pluripotent stem cell-derived neural crest cells for corneal endothelial regeneration
Abstract
Background: The corneal endothelium maintains corneal hydration through the barrier and pump function, while its dysfunction may cause corneal edema and vision reduction. Considering its development from neural crest cells (NCCs), here we investigated the efficacy of the human induced pluripotent stem cell (hiPSC)-derived NCCs for corneal endothelial regeneration in rabbits.
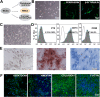
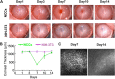
Methods: Directed differentiation of hiPSC-derived NCCs was achieved using the chemically defined medium containing GSK-3 inhibitor and TGF-β inhibitor. The differentiated cells were characterized by immunofluorescence staining, FACS analysis, and in vitro multi-lineage differentiation capacity. For in vivo functional evaluation, 1.0 × 106 hiPSC-derived NCCs or NIH-3 T3 fibroblasts (as control) combined with 100 μM Y-27632 were intracamerally injected into the anterior chamber of rabbits following removal of corneal endothelium. Rabbit corneal thickness and phenotype changes of the transplanted cells were examined at 7 and 14 days with handy pachymeter, dual-immunofluorescence staining, and quantitative RT-PCR.
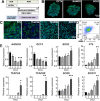
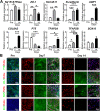
Results: The hiPSC-derived NCCs were differentiated homogenously through 7 days of induction and exhibited multi-lineage differentiation capacity into peripheral neurons, mesenchymal stem cells, and corneal keratocytes. After 7 days of intracameral injection in rabbit, the hiPSC-derived NCCs led to a gradual recovery of normal corneal thickness and clarity, when comparing to control rabbit with fibroblasts injection. However, the recovery efficacy after 14 days deteriorated and caused the reappearance of corneal edema. Mechanistically, the transplanted cells exhibited the impaired maturation, cellular senescence, and endothelial-mesenchymal transition (EnMT) after the early stage of the in vivo directional differentiation.
Conclusions: Transplantation of the hiPSC-derived NCCs rapidly restored rabbit corneal thickness and clarity. However, the long-term recovery efficacy was impaired by the improper maturation, senescence, and EnMT of the transplanted cells.
Keywords: Corneal endothelium; Neural crest cells; Transplantation; iPSC.
Conflict of interest statement
The authors declare that they have no conflicts of interest.
Figures
Similar articles
-
Therapeutic Potency of Induced Pluripotent Stem-Cell-Derived Corneal Endothelial-like Cells for Corneal Endothelial Dysfunction.Int J Mol Sci. 2022 Dec 31;24(1):701. doi: 10.3390/ijms24010701. Int J Mol Sci. 2022. PMID: 36614165 Free PMC article.
-
In Vivo Differentiation of hESC-Derived Neural Crest Cells into Trabecular Meshwork Cells.Tissue Eng Part A. 2025 Jun 16. doi: 10.1089/ten.tea.2024.0343. Online ahead of print. Tissue Eng Part A. 2025. PMID: 40518990
-
Corneal Endothelial Regeneration Using Mesenchymal Stem Cells Derived from Human Umbilical Cord.Stem Cells Dev. 2018 Aug 15;27(16):1097-1108. doi: 10.1089/scd.2017.0297. Epub 2018 Jul 25. Stem Cells Dev. 2018. PMID: 29929442
-
[Transplantation of corneal endothelial cells].Nippon Ganka Gakkai Zasshi. 2002 Dec;106(12):805-35; discussion 836. Nippon Ganka Gakkai Zasshi. 2002. PMID: 12610838 Review. Japanese.
-
Corneal Endothelial-like Cells Derived from Induced Pluripotent Stem Cells for Cell Therapy.Int J Mol Sci. 2023 Aug 4;24(15):12433. doi: 10.3390/ijms241512433. Int J Mol Sci. 2023. PMID: 37569804 Free PMC article. Review.
Cited by
-
Focus on seed cells: stem cells in 3D bioprinting of corneal grafts.Front Bioeng Biotechnol. 2024 Jul 10;12:1423864. doi: 10.3389/fbioe.2024.1423864. eCollection 2024. Front Bioeng Biotechnol. 2024. PMID: 39050685 Free PMC article. Review.
-
Long-term corneal recovery by simultaneous delivery of hPSC-derived corneal endothelial precursors and nicotinamide.J Clin Invest. 2022 Jan 4;132(1):e146658. doi: 10.1172/JCI146658. J Clin Invest. 2022. PMID: 34981789 Free PMC article.
-
Corneal Regeneration Using Adipose-Derived Mesenchymal Stem Cells.Cells. 2022 Aug 16;11(16):2549. doi: 10.3390/cells11162549. Cells. 2022. PMID: 36010626 Free PMC article. Review.
-
RAD21 deficiency drives corneal to scleral differentiation fate switching via upregulating WNT9B.iScience. 2024 May 3;27(6):109875. doi: 10.1016/j.isci.2024.109875. eCollection 2024 Jun 21. iScience. 2024. PMID: 38774716 Free PMC article.
-
Corneal endothelial regeneration in human eyes using endothelium-free grafts.BMC Ophthalmol. 2022 Jan 21;22(1):32. doi: 10.1186/s12886-022-02260-x. BMC Ophthalmol. 2022. PMID: 35062892 Free PMC article.
References
-
- Bourne WM, Nelson LR, Hodge DO. Central corneal endothelial cell changes over a ten-year period. Invest Ophthalmol Vis Sci. 1997;38(3):779–782. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

