Splenic macrophage phagocytosis of intravenously infused mesenchymal stromal cells attenuates tumor localization
- PMID: 33781710
- PMCID: PMC8920534
- DOI: 10.1016/j.jcyt.2020.04.102
Splenic macrophage phagocytosis of intravenously infused mesenchymal stromal cells attenuates tumor localization
Abstract
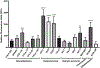
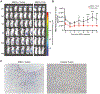
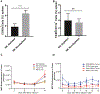
Mesenchymal stromal cells (MSCs) possess remarkable tumor tropism, making them ideal vehicles to deliver tumor-targeted therapeutic agents; however, their value in clinical medicine has yet to be realized. A barrier to clinical utilization is that only a small fraction of infused MSCs ultimately localize to the tumor. In an effort to overcome this obstacle, we sought to enhance MSC trafficking by focusing on the factors that govern MSC arrival within the tumor microenvironment. Our findings show that MSC chemoattraction is only present in select tumors, including osteosarcoma, and that the chemotactic potency among similar tumors varies substantially. Using an osteosarcoma xenograft model, we show that human MSCs traffic to the tumor within several hours of infusion. After arrival, MSCs are observed to localize in clusters near blood vessels and MSC-associated bioluminescence signal intensity is increased, suggesting that the seeded cells expand after engraftment. However, our studies reveal that a significant portion of MSCs are eliminated en route by splenic macrophage phagocytosis, effectively limiting the number of cells available for tumor engraftment. To increase MSC survival, we transiently depleted macrophages with liposomal clodronate, which resulted in increased tumor localization without substantial reduction in tumor-associated macrophages. Our data suggest that transient macrophage depletion will significantly increase the number of MSCs in the spleen and thus improve MSC localization within a tumor, theoretically increasing the effective dose of an anti-cancer agent. This strategy may subsequently improve the clinical efficacy of MSCs as vehicles for the tumor-directed delivery of therapeutic agents.
Keywords: cancer cell therapy; lentiviral transduction; mesenchymal stromal cells (MSCs); phagocytosis; splenic macrophage; stem cell transplantation; tumor homing.
Copyright © 2021. Published by Elsevier Inc.
Conflict of interest statement
Declaration of Competing Interest
The authors have no commercial, proprietary, or financial interest in the products or companies described in this article.
Figures




Similar articles
-
Mesenchymal stromal cells mediated delivery of photoactive nanoparticles inhibits osteosarcoma growth in vitro and in a murine in vivo ectopic model.J Exp Clin Cancer Res. 2020 Feb 22;39(1):40. doi: 10.1186/s13046-020-01548-4. J Exp Clin Cancer Res. 2020. PMID: 32087737 Free PMC article.
-
Macrophages at the nexus of mesenchymal stromal cell potency: The emerging role of chemokine cooperativity.Stem Cells. 2021 Sep;39(9):1145-1154. doi: 10.1002/stem.3380. Epub 2021 Apr 6. Stem Cells. 2021. PMID: 33786935 Free PMC article. Review.
-
Human umbilical cord-derived mesenchymal stem cells alleviate insulin resistance in diet-induced obese mice via an interaction with splenocytes.Stem Cell Res Ther. 2022 Mar 21;13(1):109. doi: 10.1186/s13287-022-02791-6. Stem Cell Res Ther. 2022. PMID: 35313972 Free PMC article.
-
Mesenchymal Stromal Cells Are Retained in the Porcine Renal Cortex Independently of Their Metabolic State After Renal Intra-Arterial Infusion.Stem Cells Dev. 2019 Sep 15;28(18):1224-1235. doi: 10.1089/scd.2019.0105. Epub 2019 Aug 1. Stem Cells Dev. 2019. PMID: 31280676
-
Mesenchymal stem/stromal cells as next-generation drug delivery vehicles for cancer therapeutics.Expert Opin Drug Deliv. 2021 Nov;18(11):1627-1642. doi: 10.1080/17425247.2021.1960309. Epub 2021 Aug 5. Expert Opin Drug Deliv. 2021. PMID: 34311638 Review.
Cited by
-
Pharmacokinetic characteristics of mesenchymal stem cells in translational challenges.Signal Transduct Target Ther. 2024 Sep 13;9(1):242. doi: 10.1038/s41392-024-01936-8. Signal Transduct Target Ther. 2024. PMID: 39271680 Free PMC article. Review.
-
Plasticity and crosstalk of mesenchymal stem cells and macrophages in immunomodulation in sepsis.Front Immunol. 2024 Jan 30;15:1338744. doi: 10.3389/fimmu.2024.1338744. eCollection 2024. Front Immunol. 2024. PMID: 38352879 Free PMC article. Review.
-
Secondary Lymphoid Organs in Mesenchymal Stromal Cell Therapy: More Than Just a Filter.Front Immunol. 2022 Jun 16;13:892443. doi: 10.3389/fimmu.2022.892443. eCollection 2022. Front Immunol. 2022. PMID: 35784291 Free PMC article. Review.
-
Mesenchymal Stem/Stromal Cells as a Vehicle for Cytokine Delivery: An Emerging Approach for Tumor Immunotherapy.Front Med (Lausanne). 2021 Aug 27;8:721174. doi: 10.3389/fmed.2021.721174. eCollection 2021. Front Med (Lausanne). 2021. Retraction in: Front Med (Lausanne). 2023 Sep 04;10:1285195. doi: 10.3389/fmed.2023.1285195. PMID: 34513882 Free PMC article. Retracted. Review.
-
From Interaction to Intervention: How Mesenchymal Stem Cells Affect and Target Triple-Negative Breast Cancer.Biomedicines. 2023 Apr 15;11(4):1182. doi: 10.3390/biomedicines11041182. Biomedicines. 2023. PMID: 37189800 Free PMC article. Review.
References
-
- Pittenger MF, Mackay AM, Beck SC, et al. Multilineage potential of adult human mesenchymal stem cells. Science 1999;284:143–7. - PubMed
-
- Spaeth E, Klopp A, Dembinski J, et al. Inflammation and tumor microenvironments: defining the migratory itinerary of mesenchymal stem cells. Gene therapy 2008;15:730–8. - PubMed
-
- Nakamizo A, Marini F, Amano T, et al. Human bone marrow-derived mesenchymal stem cells in the treatment of gliomas. Cancer research 2005;65:3307–18. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

