Ivermectin reduces in vivo coronavirus infection in a mouse experimental model
- PMID: 33785846
- PMCID: PMC8010049
- DOI: 10.1038/s41598-021-86679-0
Ivermectin reduces in vivo coronavirus infection in a mouse experimental model
Abstract
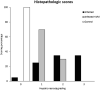
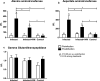
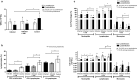
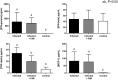
The objective of this study was to test the effectiveness of ivermectin for the treatment of mouse hepatitis virus (MHV), a type 2 family RNA coronavirus similar to SARS-CoV-2. Female BALB/cJ mice were infected with 6,000 PFU of MHV-A59 (group infected, n = 20) or infected and then immediately treated with a single dose of 500 µg/kg ivermectin (group infected + IVM, n = 20) or were not infected and treated with PBS (control group, n = 16). Five days after infection/treatment, the mice were euthanized and the tissues were sampled to assess their general health status and infection levels. Overall, the results demonstrated that viral infection induced typical MHV-caused disease, with the livers showing severe hepatocellular necrosis surrounded by a severe lymphoplasmacytic inflammatory infiltration associated with a high hepatic viral load (52,158 AU), while mice treated with ivermectin showed a better health status with a lower viral load (23,192 AU; p < 0.05), with only a few having histopathological liver damage (p < 0.05). No significant differences were found between the group infected + IVM and control group mice (P = NS). Furthermore, serum transaminase levels (aspartate aminotransferase and alanine aminotransferase) were significantly lower in the treated mice than in the infected animals. In conclusion, ivermectin diminished the MHV viral load and disease in the mice, being a useful model for further understanding this therapy against coronavirus diseases.
Conflict of interest statement
The authors declare no competing interests.
Figures





Similar articles
-
A Biosafety Level 2 Mouse Model for Studying Betacoronavirus-Induced Acute Lung Damage and Systemic Manifestations.J Virol. 2021 Oct 27;95(22):e0127621. doi: 10.1128/JVI.01276-21. Epub 2021 Sep 8. J Virol. 2021. PMID: 34495692 Free PMC article.
-
Combination Treatment With Remdesivir and Ivermectin Exerts Highly Synergistic and Potent Antiviral Activity Against Murine Coronavirus Infection.Front Cell Infect Microbiol. 2021 Jul 30;11:700502. doi: 10.3389/fcimb.2021.700502. eCollection 2021. Front Cell Infect Microbiol. 2021. PMID: 34395311 Free PMC article.
-
Expression of hemagglutinin/esterase by a mouse hepatitis virus coronavirus defective-interfering RNA alters viral pathogenesis.Virology. 1998 Mar 1;242(1):170-83. doi: 10.1006/viro.1997.8993. Virology. 1998. PMID: 9501044 Free PMC article.
-
Synergistic antiviral effect of a combination of mouse interferon-alpha and interferon-gamma on mouse hepatitis virus.J Med Virol. 2003 Feb;69(2):188-94. doi: 10.1002/jmv.10286. J Med Virol. 2003. PMID: 12683406 Free PMC article.
-
Of Mice and Men: The Coronavirus MHV and Mouse Models as a Translational Approach to Understand SARS-CoV-2.Viruses. 2020 Aug 12;12(8):880. doi: 10.3390/v12080880. Viruses. 2020. PMID: 32806708 Free PMC article. Review.
Cited by
-
Basic reproduction numbers of three strains of mouse hepatitis viruses in mice.Microbiol Immunol. 2022 Apr;66(4):166-172. doi: 10.1111/1348-0421.12961. Epub 2022 Jan 18. Microbiol Immunol. 2022. PMID: 34984727 Free PMC article.
-
Review of the Emerging Evidence Demonstrating the Efficacy of Ivermectin in the Prophylaxis and Treatment of COVID-19.Am J Ther. 2021 Apr 22;28(3):e299-e318. doi: 10.1097/MJT.0000000000001377. Am J Ther. 2021. PMID: 34375047 Free PMC article. Review.
-
Interaction of Epigallocatechin Gallate and Quercetin with Spike Glycoprotein (S-Glycoprotein) of SARS-CoV-2: In Silico Study.Biomedicines. 2022 Nov 29;10(12):3074. doi: 10.3390/biomedicines10123074. Biomedicines. 2022. PMID: 36551830 Free PMC article.
-
Antiparasitic Drugs against SARS-CoV-2: A Comprehensive Literature Survey.Microorganisms. 2022 Jun 24;10(7):1284. doi: 10.3390/microorganisms10071284. Microorganisms. 2022. PMID: 35889004 Free PMC article. Review.
-
Ivermectin-Induced Acute Psychosis in Patients Infected With COVID-19 Pneumonia.Cureus. 2022 Jun 21;14(6):e26141. doi: 10.7759/cureus.26141. eCollection 2022 Jun. Cureus. 2022. PMID: 35747110 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

