Ferulic Acid Ameliorates Alzheimer's Disease-like Pathology and Repairs Cognitive Decline by Preventing Capillary Hypofunction in APP/PS1 Mice
- PMID: 33786807
- PMCID: PMC8423929
- DOI: 10.1007/s13311-021-01024-7
Ferulic Acid Ameliorates Alzheimer's Disease-like Pathology and Repairs Cognitive Decline by Preventing Capillary Hypofunction in APP/PS1 Mice
Abstract
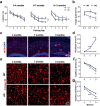
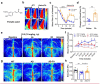
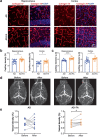
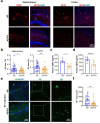
Brain capillaries are crucial for cognitive functions by supplying oxygen and other nutrients to and removing metabolic wastes from the brain. Recent studies have demonstrated that constriction of brain capillaries is triggered by beta-amyloid (Aβ) oligomers via endothelin-1 (ET1)-mediated action on the ET1 receptor A (ETRA), potentially exacerbating Aβ plaque deposition, the primary pathophysiology of Alzheimer's disease (AD). However, direct evidence is still lacking whether changes in brain capillaries are causally involved in the pathophysiology of AD. Using APP/PS1 mouse model of AD (AD mice) relative to age-matched negative littermates, we identified that reductions of density and diameter of hippocampal capillaries occurred from 4 to 7 months old while Aβ plaque deposition and spatial memory deficit developed at 7 months old. Notably, the injection of ET1 into the hippocampus induced early Aβ plaque deposition at 5 months old in AD mice. Conversely, treatment of ferulic acid against the ETRA to counteract the ET1-mediated vasoconstriction for 30 days prevented reductions of density and diameter of hippocampal capillaries as well as ameliorated Aβ plaque deposition and spatial memory deficit at 7 months old in AD mice. Thus, these data suggest that reductions of density and diameter of hippocampal capillaries are crucial for initiating Aβ plaque deposition and spatial memory deficit at the early stages, implicating the development of new therapies for halting or curing memory decline in AD.
Keywords: APP/PS1 mouse; Alzheimer’s disease; Aβ plaque; Endothelin-1; Ferulic acid (FA); Hippocampus.
© 2021. The Author(s).
Conflict of interest statement
Some authors are listed as the inventors of a CN Patent (application no. 202010395578.7). The other authors declare that they have no competing interests.
Figures


References
-
- Beyreuther K, Masters CL. Amyloid precursor protein (APP) and beta A4 amyloid in the etiology of Alzheimer’s disease: precursor-product relationships in the derangement of neuronal function. Brain pathology (Zurich, Switzerland). 1991;1(4):241–251. doi: 10.1111/j.1750-3639.1991.tb00667.x. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

