Toward a Systematic Nomenclature for (Neo)Lignanamides
- PMID: 33787264
- PMCID: PMC8155391
- DOI: 10.1021/acs.jnatprod.0c00792
Toward a Systematic Nomenclature for (Neo)Lignanamides
Abstract
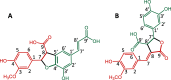
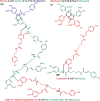
Phenylalkenoic acid amides, often referred to as phenol amides or hydroxycinnamic acid amides, are bioactive phytochemicals, whose bioactivity can be enhanced by coupling to form dimers or oligomers. Phenylalkenoic acid amides consist of a (hydroxy)cinnamic acid derivative (i.e., the phenylalkenoic acid subunit) linked to an amine-containing compound (i.e., the amine subunit) via an amide bond. The phenylalkenoic acid moiety can undergo oxidative coupling, either catalyzed by oxidative enzymes or due to autoxidation, which leads to the formation of (neo)lignanamides. Dimers described in the literature are often named after the species in which the compound was first discovered; however, the naming of these compounds lacks a systematic approach. We propose a new nomenclature, inspired by the existing system used for hydroxycinnamic acid dimers and lignin. In the proposed systematic nomenclature for (neo)lignanamides, compound names will be composed of three-letter codes and prefixes denoting the subunits, and numbers that indicate the carbon atoms involved in the linkage between the monomeric precursors. The proposed nomenclature is consistent, future-proof, and systematic.
Conflict of interest statement
The authors declare no competing financial interest.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

