Fa(c)t checking: How fatty acids shape metabolism and function of macrophages and dendritic cells
- PMID: 33788250
- PMCID: PMC8359938
- DOI: 10.1002/eji.202048944
Fa(c)t checking: How fatty acids shape metabolism and function of macrophages and dendritic cells
Abstract
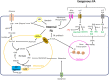
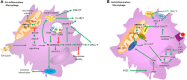
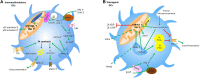
In recent years there have been major advances in our understanding of the role of free fatty acids (FAs) and their metabolism in shaping the functional properties of macrophages and DCs. This review presents the most recent insights into how cell intrinsic FA metabolism controls DC and macrophage function, as well as the current evidence of the importance of various exogenous FAs (such as polyunsaturated FAs and their oxidation products-prostaglandins, leukotrienes, and proresolving lipid mediators) in affecting DC and macrophage biology, by modulating their metabolic properties. Finally, we explore whether targeted modulation of FA metabolism of myeloid cells to steer their function could hold promise in therapeutic settings.
Keywords: Dendritic cells; Fatty Acids; Macrophages; Metabolism; Proresolving mediators.
© 2021 The Authors. European Journal of Immunology published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no financial or commercial conflict of interest.
Figures
Similar articles
-
De Novo Fatty Acid Synthesis During Mycobacterial Infection Is a Prerequisite for the Function of Highly Proliferative T Cells, But Not for Dendritic Cells or Macrophages.Front Immunol. 2018 Apr 5;9:495. doi: 10.3389/fimmu.2018.00495. eCollection 2018. Front Immunol. 2018. PMID: 29675017 Free PMC article.
-
Inhibition of fatty acid metabolism by etomoxir or TOFA suppresses murine dendritic cell activation without affecting viability.Immunopharmacol Immunotoxicol. 2019 Jun;41(3):361-369. doi: 10.1080/08923973.2019.1616754. Epub 2019 Jun 2. Immunopharmacol Immunotoxicol. 2019. PMID: 31155984 Free PMC article.
-
Human monocyte-derived dendritic cells turn into foamy dendritic cells with IL-17A.J Lipid Res. 2015 Jun;56(6):1110-22. doi: 10.1194/jlr.M054874. Epub 2015 Apr 1. J Lipid Res. 2015. PMID: 25833686 Free PMC article.
-
Lipid metabolism in dendritic cell biology.Immunol Rev. 2023 Aug;317(1):137-151. doi: 10.1111/imr.13215. Epub 2023 May 12. Immunol Rev. 2023. PMID: 37172120 Free PMC article. Review.
-
Fatty acid metabolism in macrophages: a target in cardio-metabolic diseases.Curr Opin Lipidol. 2017 Feb;28(1):19-26. doi: 10.1097/MOL.0000000000000370. Curr Opin Lipidol. 2017. PMID: 27870652 Review.
Cited by
-
Live-Cell Imaging of Sterculic Acid-a Naturally Occurring 1,2-Cyclopropene Fatty Acid-by Bioorthogonal Reaction with Turn-On Tetrazine-Fluorophore Conjugates.Angew Chem Int Ed Engl. 2022 Sep 19;61(38):e202207640. doi: 10.1002/anie.202207640. Epub 2022 Aug 8. Angew Chem Int Ed Engl. 2022. PMID: 35838324 Free PMC article.
-
From Skin and Gut to the Brain: The Infectious Journey of the Human Commensal Fungus Malassezia and Its Neurological Consequences.Mol Neurobiol. 2025 Jan;62(1):533-556. doi: 10.1007/s12035-024-04270-w. Epub 2024 Jun 14. Mol Neurobiol. 2025. PMID: 38871941 Review.
-
Host Lipid Manipulation by Intracellular Bacteria: Moonlighting for Immune Evasion.J Membr Biol. 2023 Dec;256(4-6):393-411. doi: 10.1007/s00232-023-00296-8. Epub 2023 Nov 8. J Membr Biol. 2023. PMID: 37938349 Review.
-
Targeting Strategies for Aberrant Lipid Metabolism Reprogramming and the Immune Microenvironment in Esophageal Cancer: A Review.J Oncol. 2022 Sep 5;2022:4257359. doi: 10.1155/2022/4257359. eCollection 2022. J Oncol. 2022. PMID: 36106333 Free PMC article. Review.
-
Cryptotanshinone attenuates LPS-induced acute lung injury by regulating metabolic reprogramming of macrophage.Front Med (Lausanne). 2023 Jan 13;9:1075465. doi: 10.3389/fmed.2022.1075465. eCollection 2022. Front Med (Lausanne). 2023. PMID: 36714100 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

