Classical complement and inflammasome activation converge in CD14highCD16- monocytes in HIV associated TB-immune reconstitution inflammatory syndrome
- PMID: 33788899
- PMCID: PMC8041190
- DOI: 10.1371/journal.ppat.1009435
Classical complement and inflammasome activation converge in CD14highCD16- monocytes in HIV associated TB-immune reconstitution inflammatory syndrome
Abstract
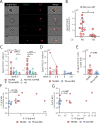
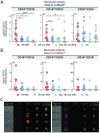
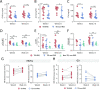
Inflammasome-derived cytokines, IL-1β and IL-18, and complement cascade have been independently implicated in the pathogenesis of tuberculosis (TB)-immune reconstitution inflammatory syndrome (TB-IRIS), a complication affecting HIV+ individuals starting antiretroviral therapy (ART). Although sublytic deposition of the membrane attack complex (MAC) has been shown to promote NLRP3 inflammasome activation, it is unknown whether these pathways may cooperatively contribute to TB-IRIS. To evaluate the activation of inflammasome, peripheral blood mononuclear cells (PBMCs) from HIV-TB co-infected patients prior to ART and at the IRIS or equivalent timepoint were incubated with a probe used to assess active caspase-1/4/5 followed by screening of ASC (apoptosis-associated speck-like protein containing a CARD domain) specks as a readout of inflammasome activation by imaging flow cytometry. We found higher numbers of monocytes showing spontaneous caspase-1/4/5+ASC-speck formation in TB-IRIS compared to TB non-IRIS patients. Moreover, numbers of caspase-1/4/5+ASC-speck+ monocytes positively correlated with IL-1β/IL-18 plasma levels. Besides increased systemic levels of C1q and C5a, TB-IRIS patients also showed elevated C1q and C3 deposition on monocyte cell surface, suggesting aberrant classical complement activation. A clustering tSNE analysis revealed TB-IRIS patients are enriched in a CD14highCD16- monocyte population that undergoes MAC deposition and caspase-1/4/5 activation compared to TB non-IRIS patients, suggesting complement-associated inflammasome activation during IRIS events. Accordingly, PBMCs from patients were more sensitive to ex-vivo complement-mediated IL-1β secretion than healthy control cells in a NLRP3-dependent manner. Therefore, our data suggest complement-associated inflammasome activation may fuel the dysregulated TB-IRIS systemic inflammatory cascade and targeting this pathway may represent a novel therapeutic approach for IRIS or related inflammatory syndromes.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



References
-
- Castelnuovo B, Manabe YC, Kiragga A, Kamya M, Easterbrook P, Kambugu A. Cause-specific mortality and the contribution of immune reconstitution inflammatory syndrome in the first 3 years after antiretroviral therapy initiation in an urban African cohort. Clin Infect Dis. 2009;49(6):965–72. 10.1086/605500 . - DOI - PubMed
-
- Hoyo-Ulloa I, Belaunzaran-Zamudio PF, Crabtree-Ramirez B, Galindo-Fraga A, Perez-Aguinaga ME, Sierra-Madero JG. Impact of the immune reconstitution inflammatory syndrome (IRIS) on mortality and morbidity in HIV-infected patients in Mexico. Int J Infect Dis. 2011;15(6):e408–14. 10.1016/j.ijid.2011.02.007 - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

