Inhibition of the Axl pathway impairs breast and prostate cancer metastasis to the bones and bone remodeling
- PMID: 33791875
- PMCID: PMC8179919
- DOI: 10.1007/s10585-021-10093-z
Inhibition of the Axl pathway impairs breast and prostate cancer metastasis to the bones and bone remodeling
Abstract
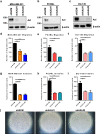
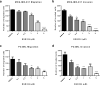
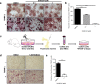
Approximately 90% of cancer-related deaths result from cancer metastasis. In prostate and breast cancers, bone is the most common site of cancer cell dissemination. Key steps in the metastatic cascade are promoted through upregulation of critical cell signaling pathways in neoplastic cells. The present study assessed the role of the receptor tyrosine kinase Axl in prostate and breast cancer cell metastasis to bones using (i) Axl knockdown neoplastic cells and osteoclast progenitor cells in vitro, (ii) intracardiac injection of Axl knockdown tumor cells in vivo, and (iii) selective Axl inhibitor BGB324. Axl inhibition in neoplastic cells significantly decreased their metastatic potential, and suppression of Axl signaling in osteoclast precursor cells also reduced the formation of mature osteoclasts. In vivo, Axl knockdown in prostate and breast cancer cells significantly suppressed the formation and progression of bone metastases. Hence, therapeutic targeting of Axl may impair tumor metastasis to the bones through neoplastic and host cell signaling axes.
Keywords: Axl; Bone metastasis; Osteoclastogenesis; Receptor tyrosine kinase.
Conflict of interest statement
The authors declare no conflict of interest/competing interest.
Figures



Similar articles
-
Hypoxia stabilizes GAS6/Axl signaling in metastatic prostate cancer.Mol Cancer Res. 2012 Jun;10(6):703-12. doi: 10.1158/1541-7786.MCR-11-0569. Epub 2012 Apr 19. Mol Cancer Res. 2012. PMID: 22516347 Free PMC article.
-
The Receptor Tyrosine Kinase AXL Is Required at Multiple Steps of the Metastatic Cascade during HER2-Positive Breast Cancer Progression.Cell Rep. 2018 May 1;23(5):1476-1490. doi: 10.1016/j.celrep.2018.04.019. Cell Rep. 2018. PMID: 29719259
-
AXL Is a Putative Tumor Suppressor and Dormancy Regulator in Prostate Cancer.Mol Cancer Res. 2019 Feb;17(2):356-369. doi: 10.1158/1541-7786.MCR-18-0718. Epub 2018 Oct 5. Mol Cancer Res. 2019. PMID: 30291220 Free PMC article.
-
The receptor tyrosine kinase Axl in cancer: biological functions and therapeutic implications.Int J Cancer. 2014 Mar 1;134(5):1024-33. doi: 10.1002/ijc.28246. Epub 2013 Jun 4. Int J Cancer. 2014. PMID: 23649974 Review.
-
AXL kinase as a novel target for cancer therapy.Oncotarget. 2014 Oct 30;5(20):9546-63. doi: 10.18632/oncotarget.2542. Oncotarget. 2014. PMID: 25337673 Free PMC article. Review.
Cited by
-
Bone Marrow Endothelial Cells Increase Prostate Cancer Cell Apoptosis in 3D Triculture Model of Reactive Stroma.Biology (Basel). 2022 Aug 26;11(9):1271. doi: 10.3390/biology11091271. Biology (Basel). 2022. PMID: 36138750 Free PMC article.
-
Current methods for studying metastatic potential of tumor cells.Cancer Cell Int. 2022 Dec 9;22(1):394. doi: 10.1186/s12935-022-02801-w. Cancer Cell Int. 2022. PMID: 36494720 Free PMC article. Review.
-
Inhibition of Mertk Signaling Enhances Bone Healing after Tooth Extraction.J Dent Res. 2023 Sep;102(10):1131-1140. doi: 10.1177/00220345231177996. Epub 2023 Jun 22. J Dent Res. 2023. PMID: 37350025 Free PMC article.
-
Structure-based drug discovery of novel fused-pyrazolone carboxamide derivatives as potent and selective AXL inhibitors.Acta Pharm Sin B. 2023 Dec;13(12):4918-4933. doi: 10.1016/j.apsb.2023.10.002. Epub 2023 Oct 10. Acta Pharm Sin B. 2023. PMID: 38045061 Free PMC article.
-
Functional Interrogation of Ca2+ Signals in Human Cancer Cells In Vitro and Ex Vivo by Fluorescent Microscopy and Molecular Tools.Methods Mol Biol. 2023;2679:95-125. doi: 10.1007/978-1-0716-3271-0_7. Methods Mol Biol. 2023. PMID: 37300611
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

