Inducible phospholipid transfer protein deficiency ameliorates atherosclerosis
- PMID: 33798923
- PMCID: PMC8096707
- DOI: 10.1016/j.atherosclerosis.2021.03.011
Inducible phospholipid transfer protein deficiency ameliorates atherosclerosis
Abstract
Background and aims: Atherosclerosis progression and regression studies are related to its prevention and treatment. Although we have gained extensive knowledge on germline phospholipid transfer protein (PLTP) deficiency, the effect of inducible PLTP deficiency in atherosclerosis remains unexplored.
Methods: We generated inducible PLTP (iPLTP)-knockout (KO) mice and measured their plasma lipid levels after feeding a normal chow or a Western-type diet. Adenovirus associated virus-proprotein convertase subtilisin/kexin type 9 (AAV-PCSK9) was used to induce hypercholesterolemia in the mice. Collars were placed around the common carotid arteries, and atherosclerosis progression and regression in the carotid arteries and aortic roots were evaluated.
Results: On a normal chow diet, iPLTP-KO mice exhibited decreased cholesterol, phospholipid, apoA-I, and apoB levels compared with control mice. Furthermore, the overall amount of high-density lipoprotein (HDL) particles was reduced in these mice, but this effect was more profound for larger HDL particles. On a Western-type diet, iPLTP-KO mice again exhibited reduced levels of all tested lipids, even though the basal lipid levels were increased. Additionally, these mice displayed significantly reduced atherosclerotic plaque sizes with increased plaque stability. Importantly, inducible PLTP deficiency significantly ameliorated atherosclerosis by reducing the size of established plaques and the number of macrophages in the plaques without causing lipid accumulation in the liver.
Conclusions: Induced PLTP deficiency in adult mice reduces plasma total cholesterol and triglycerides, prevents atherosclerosis progression, and promotes atherosclerosis regression. Thus, PLTP inhibition is a promising therapeutic approach for atherosclerosis.
Keywords: Atherosclerosis progression and regression; Inducible PLTP gene knockout mice; Lipoprotein metabolism; Stability of atherosclerotic plaque.
Copyright © 2021 Elsevier B.V. All rights reserved.
Figures
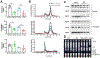
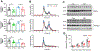



Comment in
-
Atherosclerosis regression is associated with both infiltration of new leukocytes into the plaque and a shift in macrophage polarization towards a more migratory phenotype.Atherosclerosis. 2022 Sep;356:50-52. doi: 10.1016/j.atherosclerosis.2022.06.1013. Epub 2022 Jun 18. Atherosclerosis. 2022. PMID: 35753822 No abstract available.
-
PLTP deficiency-mediated atherosclerosis regression could be related to sphinogosine-1-phosphate reduction.Atherosclerosis. 2022 Sep;356:53-55. doi: 10.1016/j.atherosclerosis.2022.07.007. Epub 2022 Jul 15. Atherosclerosis. 2022. PMID: 35940962 Free PMC article. No abstract available.
References
-
- Tall AR, Krumholz S, Olivecrona T, Deckelbaum RJ, Plasma phospholipid transfer protein enhances transfer and exchange of phospholipids between very low density lipoproteins and high density lipoproteins during lipolysis, J. Lipid Res 26 (1985) 842–851. - PubMed
-
- Jauhiainen M, Metso J, Pahlman R, Blomqvist S, van Tol A, Ehnholm C, Human plasma phospholipid transfer protein causes high density lipoprotein conversion, J. Biol. Chem 268 (1993) 4032–4036. - PubMed
-
- Schlitt A, Bickel C, Thumma P, Blankenberg S, Rupprecht HJ, Meyer J, Jiang XC, High plasma phospholipid transfer protein levels as a risk factor for coronary artery disease, Arterioscler. Thromb. Vasc. Biol 23 (2003) 1857–1862. - PubMed
-
- Vergeer M, Boekholdt SM, Sandhu MS, Ricketts SL, Wareham NJ, Brown MJ, de Faire U, Leander K, Gigante B, Kavousi M, Hofman A, Uitterlinden AG, van Duijn CM, Witteman JC, Jukema JW, Schadt EE, van der Schoot E, Kastelein JJ, Khaw KT, Dullaart RP, van Tol A, Trip MD, Dallinga-Thie GM, Genetic variation at the phospholipid transfer protein locus affects its activity and high-density lipoprotein size and is a novel marker of cardiovascular disease susceptibility, Circulation 122 (2010) 470–477. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

